About 58 million people worldwide are infected with chronic Hepatitis C Virus (HCV). An estimated 290,000 of these carriers die yearly from liver cirrhosis and primary liver cancer, mostly because they are unaware of being infected or unaware of their status.1 This means that the WHO goal of eliminating HCV by 2030 faces a considerable challenge. Despite the availability and success of short-course, highly effective Direct Acting Antivirals (DAAs) with few side effects and recommendation for a treat-all approach.2 Although this strategy for HCV elimination is cost-effective and cost-saving, most low- and middle-income countries (LMICs) with a considerable HCV burden, like Cameroon, are currently not investing in elimination.3
Recently, sharp drops in DAA prices have been reached through tiered pricing, which uses a country’s national income and voluntary license agreements to determine prices.2 The government of Cameroon – a Central African country with about 28 million inhabitants and a high HCV prevalence, especially among those aged 45 and above – also negotiated considerably lower prices for both branded and generic WHO-pre-qualified DAA. However, because of a lack of healthcare insurance, patients are forced to pay out-of-pocket for the DAA medication, diagnostics and specialist consultations. The combined costs are unaffordable for most chronic HCV carriers in Cameroon. Another barrier to early treatment is the lack of screening programs to detect asymptomatic HCV patients. Having showed high treatment success rates for HCV with DAAs in clinical settings in Cameroon,4 accessibility and provision of HCV care continuum should subsequently be addressed to ensure effective large-scale implementation and potential elimination.
The unaffordability of DAA treatment for most chronic HCV carriers in Sub-Saharan Africa encouraged policymakers and scientists to develop innovative financing mechanisms. Many African public health systems fail to deliver efficient healthcare, partly due to a lack of (tax-based) resources, forcing many patients to pay out of pocket for the health services they use. While investing in the healthcare supply chain is often considered too risky from a private investment point of view, other areas of collaboration between the government and the private sector – so-called public-private partnerships (PPP) - increasingly happen in the healthcare sector. In many LMICs, the gap between healthcare needs and healthcare provision results from poor medicine supply, scarce resources, and weak healthcare infrastructures. Hence, PPP can enable vulnerable populations to access health care5 because it increases the budget available within the health sector.
A Development Impact Bond (DIB) is an innovative financing mechanism in which private investors provide pre-payment of development program expenses. At the same time, public agencies or donors repay the investor’s investment with a reasonable interest rate if the program succeeds in delivering independently measurable results that are contractually agreed upon. A DIB can especially be used in healthcare when the results of a given intervention are unequivocally measurable through a qualified third party and where there are potential governance issues. DIBs have attracted considerable interest, but the evidence base is limited.6
DIBs generally involve six agents, as displayed in Figure 1.7 The investor provides the initial funding for the project, which is directed to the intermediary. The intermediary uses this money to pay service providers interacting with the target population. The independent evaluator will validate reported outcomes once the target population is reached and treated. The outcome payer only disburses funds to the investor based on achieved successes as reported by the evaluator.
DIBs are relatively new. Currently there are 16 DIBs active worldwide, of which most are implemented in Africa in social welfare, employment and health.8 The outcome-payer can either be the government or a donor. Four preconditions generally characterize a DIB: i) there is a measurable outcome that can be validated by an independent assessor ii) there is a reasonable time horizon to achieve the desired outcomes, iii) there is evidence that the outcomes can be achieved successfully, and iv) appropriate legal and political conditions are in place to support the bond.9 The fourth condition is most important when the government is the outcome payer, while the third condition is crucial to motivate investors to provide the pre-investment.
The reported high DAA cure rate of more than 95% over a short treatment period of 12 weeks for chronic HCV carriers suggests that a DIB might be a fitting route to generate investments for treatment programs. Hence, investing in a DIB for HCV diagnosis and treatment is likely to be an attractive way of investing for private (social) investors. Cameroon is one of the first sub-Saharan countries with a reported high cure rate (>96%) with DAA. Meanwhile, the national HCV strategy to determine HCV prevalence in Cameroon is not sufficiently advanced to permit an immediate implementation of a nationwide diagnosis and treatment program. Therefore, we aim to assess the feasibility of a DIB for HCV diagnosis and treatment in the Cameroonian setting by evaluating the execution, financial and medical impact at a small-scale. This will help identify barriers and facilitators for future DIBs and generate concrete insights regarding future upscaling.
METHODS
Study design
We designed a prospective longitudinal study. The patient enrollment period was six months, and data (on treatment status and demographic characteristics of each patient) was collected at the following time points: i) at the start (t = 0), ii) after the treatment period (t = 12 weeks) and iii) after the 12 weeks waiting period before final testing for cure (t = 24 weeks). Individuals who were HCV positive and did not recover after receiving standard treatment, received second-line treatment. Data was collected at t = 36 weeks, and additional testing for cure took place at t = 48 weeks. The standard treatment for HCV-positive patients is a fixed dose of generic pangenotypic DAA (Sofosbuvir and Velpatasvir), second-line treatment is carried out with branded Sofosbuvir/Velpatasvir/Voxilaprevir. Both treatments are funded through the DIB.
Study setting & study population
The study was carried out in Yaoundé, the capital of Cameroon in Central Africa. Participants for this study are identified through blood banks because they do standard testing of potential blood donors for HCV antibodies to avoid transmission to patients. In the Cameroonian system, those who test positive are refused as blood donors, informed about being antibody HCV positive and advised to visit a medical doctor. For this study, those who were antibody HCV positive are referred to program clinics and assessed for eligibility for HCV treatment. As most antibody HCV-positive potential blood donors did not show up at the assigned clinics, we decided also to include HCV-infected persons identified by private or public Health Centers in Yaoundé. HCV-infected persons identified as eligible and willing to participate are treated in five clinics (University Teaching Hospital of Yaoundé, Essos Medical Center, Centre Medical la Cathedral, Central Hospital of Yaoundé and General Hospital of Yaoundé).
Sample size justification
Based on the financial investment in the DIB, this study intended to treat 300 patients. Based on the rate of HCV-positive persons identified per year in the blood banks, the estimation of the recruitment would be completed within six months. The target was to identify around 500 antibody-HCV positive donors to enroll the planned 300 HCV-infected persons. Of the 500 antibody-HCV positive donors, we assumed that 25% would have cleared the virus by themselves (i.e. antibody-HCV positive and HCV-RNA negative),10 10% would be false positive (first anti-HCV testing in blood bank positive but not confirmed in second test) and about 10% will be lost to follow-up. The final two assumptions are based on experts’ consultations.
Development Impact Bond design
Table 1 contains characteristics of the financing mechanism. The DIB is a revolving credit facility funded by the investor (Joep Lange Institute – a Dutch NGO, www.joeplangeinstitute.org). The investor bore the social and operational risk of project success in exchange for a basic interest rate of 5% per annum and a 1% facility fee. The fund’s Facility Agent, Global Loan Agency Services (GLAS), administered the loan and managed the cash flows. Success is defined as the number of chronic HCV carriers who are HCV-RNA negative at t = 24 weeks (12 weeks after completion of DAA treatment). The Agence Nationale de Recherche sur le Sida (ANRS) in Cameroon, an internationally recognized medical research and monitoring organization, acts as a validating agency and verifies the cured patient data. The outcome is dichotomous: cured (HCV-RNA negative) or not cured (HCV-RNA positive). Once validated, fixed outcome payments per cured patient are made by the outcome payer (Achmea Foundation: www.achmea.nl/foundation) to the investor. The investor earns bonus interest from the outcome payor of 3% per annum if the cure rate is 90% or more. The staff providing HCV treatment to the target population, including doctors, nurses, pharmacists, and the coordination team, are eligible to receive a bonus of up to 10% of their annual fee. The team determines the amount paid to lead based on their performance, identified as patient satisfaction and quality of data administration. The staff knew they could receive a bonus, but the amount was not communicated beforehand. The target population, HCV chronic carriers, were aware they were treated as part of the study and provided written informed consent.
Cost estimations
The treatment and diagnostic costs per patient were estimated through a financial model. We considered the expected enrollment, adherence, and cure rates in this model. The costs were projected based on experience from the previous phase of this study, and quotations were negotiated for service and commodity providers. Cameroonian institutions that participated in the project (e.g. blood banks and treatment centers) were paid a fixed monthly amount for periods during which they were active. Fixed unit costs were agreed upon with the Centre Pasteur du Cameroon for diagnostic and analytical services, although principal reagents for viral load testing were sourced directly from the supplier. Prices largely remained fixed for the duration of the project, with only minor changes that reflected changing market conditions. A 5% cost contingency allowance was included in the DIB costing to ensure adequate funding. The costs for the legal services to formalize all required legal agreements, as well as program management and oversight by the intermediary, were covered in-kind outside of the DIB. A summary of the estimated cost breakdown based on the financial model – a scenario prediction and the actual costs - can be found in the Online Supplementary Document, appendix 1.
We requested a patient contribution (CFA 50.000 / €76), less than one-fourth of the negotiated price of the treatment medication, to ensure buy-in and to further motivate the patients to adhere to treatment. If patients were unable to pay their contribution, they were not excluded from the study. Based on the patient contribution and overall forecast costs, we defined the funds necessary from the investor to finance the project fully. Cost transparency was needed to determine the size of the cash-flows (the arrows in Figure 1). The investor (Joep Lange Institute) committed a short-term, revolving investment of up to a maximum outstanding of €230,000. This commitment was based on the estimated maximum outstanding investment per the financial model underlying the DIB. The maximum potential outcome payment for the outcome payer (Achmea Foundation) was planned at €397,800 based on outcome payments of €1,326 per cured patient, to a maximum of 300 cured patients. The outcome payments were expected to fully reimburse the investor and cover the basic and bonus interest on the investment and fee to use the facility. Funding from the Investor and the Outcome Payer were in EUR as the funding of the program originated from Europe. The exchange rate between the Euro and the Cameroonian franc (CFA) is fixed at 1 Euro to 656 CFA.
Evaluation framework
This evaluation study is guided by the six agents model depicted in Figure 1. In our setting, an experimental design with an intervention and comparable control group is impossible; thus, we cannot claim to estimate effects. However, we systemically evaluated the DIB using the proposed framework (see Online Supplementary Document, appendix 2). This framework is organized around a theory of change showing how inputs into DIBs lead to processes expected to produce impacts. The evaluation consists of qualitative and quantitative components.
Qualitative component
We conducted stakeholder interviews and thus applied a purposive sampling strategy. Relevant actors were identified based on the six-agents model. In addition, we identified relevant contextual actors (the Cameroonian government) and experts in the field of health financing to improve representation of all stakeholders. The stakeholder interviews were guided by the framework presented in the Online Supplementary Document, appendix 2. Hence, we adopted a framework method for the analyses of the data. Interviews were conducted both in-person and online. The interviews took place in a neutral environment where the respondents felt safe and comfortable. Before the start of the interviews, the purpose of the interview was explained and respondents were asked to read and sign the informed consent form. All interviews were recorded and transcribed. The transcripts were coded and interpreted in the qualitative analysis software NVivo by two researchers. The six assumptions and the four processes set out in the framework (see Online Supplementary Document, appendix 2) were applied in the thematic coding analyses of the transcripts. Data saturation was not considered as the primary goal, as we intentionally wanted to capture the breadth of the experiences and opinions of the stakeholders.
Quantitative component
We visualized the entire enrollment and treatment procedure to generate insights into the loss to follow-up, drop-out and cure rates. Data collection on demographic characteristics of those who start treatment include: gender, age, marital status, employment status, poverty probability index (PPI) score11 and HIV-status. The Poverty Probability Index (PPI) tool consists of ten low-cost questions developed as part of Cameroon’s 2014 Household Survey to estimate the probability that a household has a consumption below a given poverty line. The PPI score can theoretically range between 0 and 100, with 0 indicating the highest poverty likelihood and 100 the lowest. The data on the demographic characteristics were imported and analysed in Excel. To quantify the insights into the financial results of the DIB, we assessed the financial cash flows between the participating agents and estimated the per-patient costs. The budgeted and actual financial costs were stratified per cost type and estimated as described in the “cost estimations” section. We created an overview of the budgeted and actual costs in Excel.
Ethical approval
The study was approved by the Cameroon National Ethics Committee (2019/07/1178/CE/CNERSH/SP) and the MoH. All study staff signed a confidentiality agreement.
RESULTS
Study population
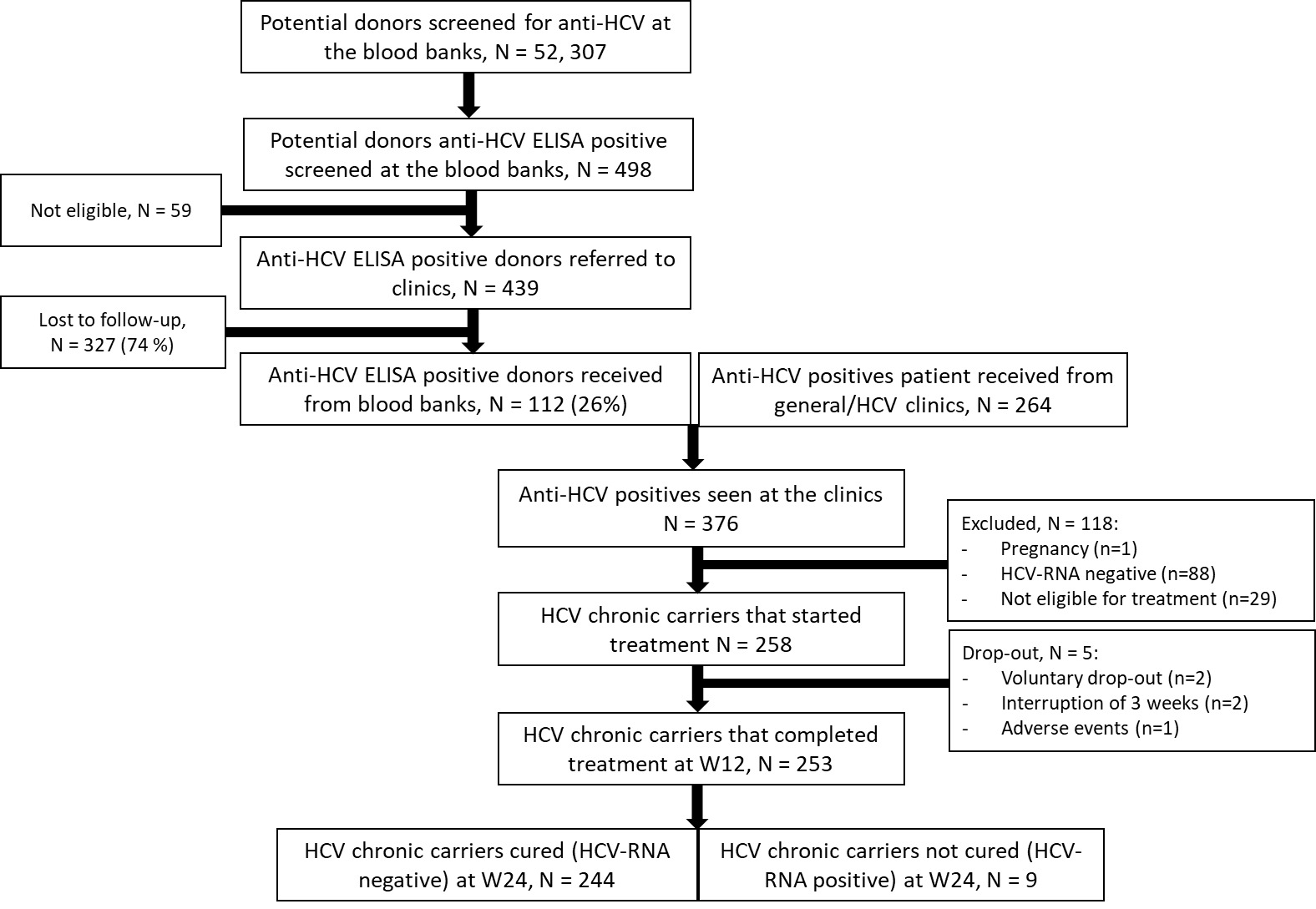
Between January 2020 and July 2021, 52,307 potential donors were screened for antibodies-HCV at eight identified blood banks around Yaoundé. From these 52,307 individuals, 439 potential donors were anti-HCV ELISA positive, informed about their test results and referred to participating clinics, of whom only 112 antibody-HCV ELISA (i.e. laboratory test) positives (26 %) were seen at the treatment clinics. Therefore, to achieve the required sample size, an additional 264 HCV-antibody positive persons identified at general clinics were recruited at treatment clinics. Of these 376 HCV-antibody positive individuals, 258 chronic HCV carriers started treatment. 118 individuals were excluded because they were HCV-RNA negative (cleared HCV infection) or not eligible for various reasons (see Figure 2). Of the 258 individuals who started treatment, 253 (98%) completed the treatment at week 12. At week 24, 244/253 (96%) are cured, and 8 out of the 9 individuals – one person refused – who were not cured at week 24 received second-line treatment for 12 weeks. All 8 were cured 12 weeks later.
Participant characteristics
Of the 258 participants who started treatment, 157 (61%) are female, and the average age is 62 (Table 2). Most (53%) are married, and a quarter are widowed. One-third of the participants are unemployed. The median PPI score is 67.5 (IQR 50 – 69.2). In addition, 4% of the study population is HIV positive.
Financial outcomes Development Impact Bond
The study was launched just before the COVID-19 pandemic and enrollment of patients was paused for six months. To continue the project, three agents – the investor, intermediary and outcome payer – agreed to invest additional money in the DIB. The total unexpected costs due to COVID-19 was €72,500 and was paid directly into the project as a once-off payment in January 2021. The unexpected costs were related to a longer recruitment period and additional staff costs. In addition, the target enrollment number was reduced from 300 to 240 because the inclusion rate was lower than anticipated.
To prove the DIB mechanism’s principle, the outcome payer (Achmea Foundation) opted to redesign the financing structure whereby delays and additional costs associated with COVID-19 (e.g., extended recruitment period) were separated from the remainder of the program. This led to splitting the patient cohort and outcome payments into two groups. Table 3 provides an overview of the results of the financial outcomes of the DIB. Group A – the initial group – includes all patients enrolled through a cut-off date set at 31 December 2021. For this group, the outcome payment per cured patient was €2,050. For the second group – Group B - the outcome payment per cured patient was reduced to €1,672. Outcome payments per cured patient were effectuated for 85 patients under Group A and 134 patients under Group B until the maximum potential outcome payment amount of €397,800 was reached. Due to this design, outcome payments were made for these 219 patients, although a further 25 patients were verified as cured, for which no outcome payment was released. This explains why the average outcome funding per treated patient - calculated as the total number of outcome payments made (€397,800) divided by the number of patients who started treatment (n=258), equaling €1,542 per patient - is lower than either Group A or B outcome payments per cured patient. The same applies to the average outcome funding per cured patient (n=244), resulting in €1,630. Additionally, the outcome payments per treated and cured patient for both Group A and B are higher than the initial outcome payment per cured patient set at €1,326 because the adjusted target enrollment number was lower (n=240) than initially planned (n=300).
The numbers presented in the paragraph above ignore the additional funding provided due to the delays and consequences of Covid-19 (€72,500) and the contributions paid by the patients. The model assumed that only 90% of the patients would pay their contribution; in reality, 256 out of the 258 patients that started treatment fully paid their contribution, making 99% and a total of €19,512. Taking these factors into account, for these amounts, the total funding provided funding for the treatment program is €489,812. The funding available per patient who started treatment (n=258) thus equals €1,898, leading to €2,007 per cured patient (n=244). The average costs per patient are based on the different cost segments making €480,315. Further results stratifying by all cost elements (e.g. medication costs, diagnostics costs) is presented in the Online Supplementary Document, appendix 1. The average cost per treated patient is € 1,858, and per cured patient is € 1,969.
Qualitative analysis
We conducted 11 interviews involving 22 persons (see the Online Supplementary Document, appendix 3). None of the approached stakeholders refused to participate. Some interviews took place in a group setting (maximum of five participants), while others had one participant. Nine interviews can be categorized within the six-agent model and the other three were contextual actors. We identified common themes throughout the manuscripts and presented these separately for the six agents and the contextual actors below.
The six-agents
Relationship between the agents
During the set-up of the DIB, the investor, the intermediary and the outcome payor were closely in contact with each other to develop and sign the legal contracts among those parties. The three-party contract stipulated that repayment of the facility was only to be made upon achievement of successes and provided for a revolving structure. This was a new structure for all involved parties, as most NGO’s are used to the regular way of project financing, which usually involves a donation agreement in the form of a lump sum payment. Hence, all parties had to identify their role, this led to an extended period of drafting contracts.
The service providers and the outcome evaluator had to work closely with each other. The outcome evaluator visited the laboratory once every month to verify the reported results. Laboratory workers explained that they were familiar with the procedure as a similar independent validation is also used for the Demographic and Health Survey (www.dhsprogram.com). However, the outcome evaluator noted that the payout for the achieved success did not happen in real-time due to the multiple validation steps.
“We did every month the validation. So once a month, I should collect all the codes of people for whom I received the notification from the base that they were declared as cured. …… But once every two months I received a message from the financing agency: ‘did you really validate?’, ‘can you confirm that, this and this.’” (Respondent I)
Compared to the regular way of working
The project introduced minor changes to the regular work procedures of the service providers. The service providers had to adopt a specific data-entry system (which was not part of their regular way of working) so the data could be externally validated. This system was considered as trustworthy and good.
To a certain extent, the service providers that delivered the treatment to the patients were incentivized to improve the quality of care because they would receive a bonus for good performance. Service providers that arranged all data entry and checked all systems were additionally incentivized with a small bonus when the project was successfully completed, which was motivating for them.
"I was already conditioned to focus on the good results. But now, it was more encouraging and stimulating because there was a bonus behind the work." (Respondent II)
Positive outcomes
Three main themes emerged as positive outcomes of the DIB. Firstly, the agents agreed that the DIB has led to a fairly accurate costing estimate of HCV diagnosis and treatment. These costing insights are considered innovative and valuable findings.
"We have given it a price. We can show quantitatively up to the euro what HCV diagnosis and treatment costs. [..] This gives transparency for a future outcome payer also if they only want to pay a certain part." (Respondent III)
Second, the DIB has led to advanced data systems. For a DIB to work, a transparent data system is required. This had to be developed and implemented. While it required training, and clinicians continued working with their paper files, it is also considered a step forward into electronic patient files.
“I don’t know if I can state it as an effect, but the consequence of the DIB is good availability of data.” (Respondent II)
“The electronic patient file is something new. That is not common here and I think that is a good experience, although it was sometimes difficult.” (Respondent II)
Third, it was mentioned that the DIB has generated additional medical insights as the same cure rates in Cameroon were achieved compared to those in high-income countries.
Contextual actors
A market for DIBs
A prerequisite for a DIB to be successful and sustainable is a flourishing DIB market. In the past decade, the DIB market did not grow exponentially as many hoped. According to the experts interviewed, finding a suitable performance indicator is challenging because many exogenous factors could influence the result. As highlighted by the six agents and the contextual actors, the performance indicator for HCV is straightforward and easily fits a DIB structure. The high cure rate in the Cameroonian setting is an interesting selling point for investors as this substantially reduces their financial risk. In addition, it was advocated that a DIB structure would make it economically and politically easier for a government to justify their spending.
“Politically it is easier for them [the government] to do because they can say to their people: we are paying for a 1,000 people this year to be cured for Hepatitis C, but we are only paying if they are cured. […] And economically, because you have confidence that you would be only paying for a most affordable price.” (Respondent IV)
The above quote suggests a scenario in which the government takes over the role of outcome payor. An important note is that because this DIB is a revolving fund and can be scaled up over time, multiple players can buy into this financing mechanism. Such a buy-in market can be attractive for the government so they can split costs among themselves and donors. More importantly, for donors to step into a larger scale DIB, it was argued that the support and involvement of the government is a prerequisite since HCV is a problem in the public domain.
“The funding of the State is indispensable. However, these funds are always likely to be insufficient and pose a problem of mobilization.” (Respondent IV)
Hepatitis C-virus
On the one hand, HCV as the subject of a DIB was considered appealing due to the nature of the performance indicator (short treatment period; high cure rate), but on the other hand, the number of people advocating for investments in HCV elimination is relatively small compared to other diseases (e.g., HIV/AIDS, tuberculosis and malaria).
“Hepatitis C is not so high on the priority agenda: middle-aged people get it and it is a salient disease. So even the people that should be fighting to get the treatment, don’t know they have it. But they end up with a painful death.” (Respondent V)
Thus, there are several reasons why HCV is not high on the agenda, but according to interviewees, what should be emphasized is that the explicit costs of curing people from HCV are not so high, and the opportunities are enormous. It was advocated among many interviewees that HCV has the potential to become an attractive disease to invest in because there are now clear cost estimates and an affordable medicine that almost guarantees a cure.
DISCUSSION
This study describes and evaluates the successful implementation of a small-scale DIB for HCV diagnosis and treatment in Cameroon. In total, 258 chronic HCV carriers were treated with a cure rate of 96%. The DIB showed that with an average per-patient outcome payment of only €1,542 it can treat one HCV patient. Worldwide, this is the first DIB devoted to HCV, and the results show that the context of HCV diagnosis and treatment is suitable for this innovative financing mechanism.
The first HCV DIB is closing, which marks the end of this study, while all elements are in place to benefit from the economies-at-scale principle. The costs of HCV service delivery used to be a black box, but this study generated valuable insights. We demonstrate that the cost to treat one chronic HCV carrier is €1,858 in a lower-middle-income country. However, this cost estimate can be further optimized because it will likely be substantially lower when assessed at scale. For instance, given this study’s is relatively small size, the local fixed costs were much higher. Additionally, many existing public health human resources could be used when implemented with governmental support. In combination with an accurate HCV prevalence estimate, it can be estimated how much investment is required to eliminate HCV in the Cameroonian setting. Such an ambition would fit nicely in a national strategy to eliminate HCV. Such a strategy is currently missing in Cameroon. Cameroon belongs to the countries that score very low on the HCV policy score index.12 HCV seems to lack a form of “sexiness” for donors and governments to prioritize this disease, possibly explained by the fact that the disease is mostly prevalent among middle-aged and older people13 Additionally, this is a silent disease: the course of the disease is asymptomatic for a long time resulting in many people being unaware of their infection and society not experiencing HCV as a danger.13
This study provides a strong financial case for HCV DIB. The financing structure could be appealing for governments in LMICs since it moves the upfront costs of service delivery to investors, and it eliminates the risk of paying for services that prove to be ineffective. However, it also requires the government to create an (additional) health budget in the context of ongoing dependence on donor funding. For donors, this can be an opportunity to cooperate with the government and ensure a measurable and sustainable impact of their funding. The bundling of different stakeholders, as private investors are also included, exemplifies a typical private-public-partnership. It is recognized that to succeed in disease elimination, the public health approach should include both public and private stakeholders and facilitate cost-sharing.14 When upfront costs are less of an issue, it could also be suggested that the government acts as an investor, as in that role, they are incentivized to ensure that the program is carried out adequately, consequently leading to reduced costs and increased revenues. This, however, is likely to be more useful in high-income settings due to the availability of more upfront resources.
Parallel to the DIB central in this study, two other DIBs were being implemented in the Cameroonian setting. The Cameroon Kangeroo Mother Care (KMC) DIB was launched in late 2018 and was closed after two and a half years of service delivery. Across 10 hospitals, 1,221 babies’ received quality KMC. The other Cameroonian DIB, The Cataract Bond, has a planned closing date in March 2023. By then, the target is to have carried out 18,000 cataract surgeries (in 5 years). While the Cameroon Ministry of Public Health is the outcome funder in the KMC DIB, the Cameroon Cataract Bond – like the HCV DIB – is fully funded by donors.15,16 The Cameroonian government’s general acknowledgement of a DIB market is demonstrated by establishing a separate unit within the Ministry of Public Health that coordinates performance-based financing activities. Even though the KMC DIB is currently closed, the sustainability of this DIB is captured in a follow-up plan in which collaboration with the Ministry of Public Health is key. The HCV DIB in this study was designed solely with funding from non-governmental organizations. Although there were several attempts to onboard the government in the DIB as a (partly) outcome payer, an agreement has never been made.
A large-scale HCV DIB could be implemented stepwise: the government may start with only paying a part of the outcome payment, supplemented by other payers, and if feasible, this amount can slowly be increased over time. An interesting by-catch of this mechanism would be that it can lead to increased trust in the government from the population. The government can justify their spending because they only pay a fixed amount for a cured patient. Conversely, this case is also interesting for private (social) investors as their risk of losing money is low, and they are likely to create sustainable social impact.
A striking observation in the study is the high drop-out rate. This rate refers to the group of potential blood donors who were lost after they were identified as chronic HCV carriers and thus were eligible to receive (almost) free life-saving treatment. It could be that these individuals are not aware of the seriousness of the disease as they do not have symptoms, requiring better and more intensive counselling after diagnosis. Therefore, to increase the treatment participation of chronic HCV carriers, an innovative Test-and-Treat approach is recommended where carriers are treated immediately after being diagnosed with a rapid test. Such an approach has previously proven its value in HIV treatment.17
Limitations
The following four limitations of the study need to be acknowledged. Firstly, this study does not allow us to obtain effect estimates of this DIB intervention. That would require an RCT with three arms: i) no financing mechanism, ii) the existing financing method, and ii) the DIB financing method. We could not run an experimental study with a counterfactual; therefore, we cannot claim to estimate and quantify effects. Secondly, the DIB incentivized the service providers by providing small bonuses based on good performance. It is considered a missed opportunity that no further quality incentives were incorporated into the design. Thirdly, our sample is relatively wealthy and, in that regard, not representative of the population in Cameroon. Referral from a peripheral provider to a more complex HCV treatment center automatically selects for the more affluent. However, the study sample’s composition is less concerned as the study’s objective was to assess the feasibility of a financing mechanism. It may only explain the patient contribution rate of 99%, which was likely lower with a more representative sample. Finally, for the continuity of the project, the government should have been involved from an earlier stage. This proved to be a challenge later in the project. The involvement of the government in donor-funded projects is increasingly being seen as a pillar for sustainability and continuity18 and should be considered necessary before starting similar projects.
CONCLUSIONS
Thanks to the availability of highly effective treatment, worldwide elimination of HCV is an achievable midterm target. When governments do not have sufficient resources to fund such elimination programs upfront, public-private partnerships can offer a solution. Our small-scale HCV DIB demonstrates that a DIB is a suitable approach for HCV diagnosis and treatment and, therefore, has the potential to diagnose and treat chronic HCV carriers in LMICs. An important requirement for scaling and longer-term sustainability is that governments assume (partly) the role of outcome payer from the start.
Acknowledgements
The authors wish to thank all HEP C-IMPACT participants and the participating clinics and also Laura Ciaffi and Pretty Mbouyap for the independent validation of the outcome. In addition, they would like to thank the following persons from the study team for their support: Dr. M. Kowo (Yaoundé University Teaching Hospital), Dr. Dang Babagnack (Centre Médical la Cathédrale), Dr. P. Talla (Yaoundé General Hospital), Dr. E. (Tchoumi Yaoundé Central Hospital), Dr. D. Simo Kamto (Essos Hospital Center) and Dr. R. Njouom (Pasteur Institute). We also thank Dr. Igna Bonfrer for her valuable comments on the draft manuscript.
Ethics statement
The study was approved by the Cameroon National Ethics Committee (2019/07/1178/CE/CNERSH/SP) and the MoH. All study staff signed a confidentiality agreement.
Data availability
The data of the study is available upon reasonable request.
Funding
The Achmea Foundation, PharmAccess Foundation and the Joep Lange Institute supported this work. This study was also financially supported by the Netherlands Ministry of Foreign Affairs through a multi-year institutional grant to PharmAccess Foundation.
Authorship contributions
C.M. Dieteren: Investigation, Formal analysis, Writing – Original Draft, Visualization.
A.C. Boers: Conceptualization, Methodology, Investigation, Writing – Review & Editing, Project administration.
W. Thomas: Conceptualization, Methodology.
O. Njoya: Conceptualization, Resources, Writing – Review & Editing.
R.A. Coutinho: Conceptualization, Methodology, Writing – Reviewing & Editing, Supervision
T. Mossus, F. Essomb, G. Wafeu, B. Agnouanang: Project management, reviewing
Disclosure of interest
The authors completed the ICMJE Disclosure of Interest Form (available upon request from the corresponding author) and disclose no relevant interests.
Additional material
The article contains additional information as an Online Supplementary Document.
Correspondence to:
Charlotte Dieteren
PharmAccess Foundation
Paasheuvelweg 25, 1105 BP Amsterdam
The Netherlands
[email protected]