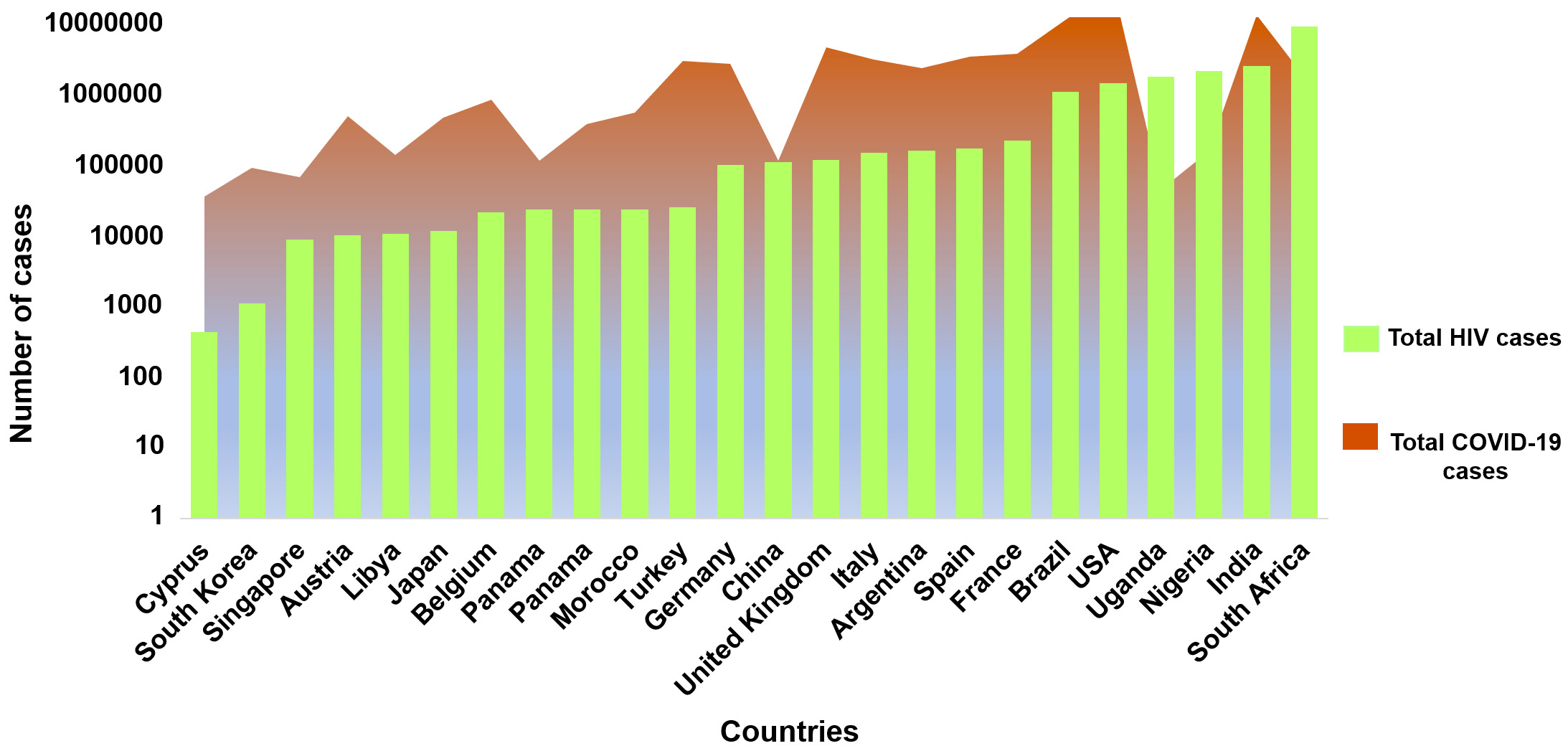
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory coronavirus 2 (SARS-CoV-2) remains a serious global public health concern. COVID-19 presentations vary from asymptomatic to severe breathlessness, which can sometimes lead to death. The overall burden of COVID-19 in society seems to have influence from other communicable diseases like human immunodeficiency virus (HIV), malaria, tuberculosis, and certain fungal and bacterial infections.1–6 To date, USA, India, and Brazil are the countries with the highest cases of COVID-19.7 Unfortunately, these countries and many others especially in sub-Saharan Africa have a significant burden of HIV 8 that will inevitably result in what Shiau et al described as HIV and SARS-CoV-2 syndemic.9 In the year 2020, ~38 million people were living with HIV and 690,000 people died due to AIDS-related illnesses globally.8 COVID-19 cases have surpassed HIV in many countries leading to concerns of an emerging syndemic. The burden of HIV is highest in South Africa followed by India and Nigeria. It is also high in the USA (Figure 1).8 India’s COVID-19 burden has been high over two waves and it has the third-highest global HIV burden with over 2 million people infected.8 To date, the largest population cohort of patients with confirmed concomitant infection of HIV and SARS-CoV-2 are reported from the United Kingdom and South Africa.10
Few anti-HIV agents may be promising drug candidates against COVID-19 and several of the multiclass combinations are already in advanced clinical trials against SARS-CoV-2.11–13 The common classes of antiretroviral therapies (ARTs) are: nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), integrase strand transfer inhibitors (INSTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), protease inhibitors (PIs), and cytochrome P4503A inhibitors (CYP3A).14 The drugs belonging to these classes are often used in different combinations depending on multiple factors involved in the HIV infection like the individual’s viral load, their T-cell count, HIV strain, the severity of the case, and other chronic illnesses.14,15 The NRTI drugs tenofovir and emtricitabine are currently in phase 3 clinical trials and may target the RNA-dependent RNA polymerase (RdRp) enzyme of SARS-CoV-2.11 RdRp may be considered as a significant druggable enzyme to be targeted in coronaviruses. RdRp is error-prone during replication that leads to the utilization of chemically modified nucleoside/nucleotide analogues as substrates instead of natural substrates.16 The chemically modified substrates cannot be processed further and hence are responsible for stalling the replication process.16 Another class of ARTs, the protease inhibitors like ritonavir, lopinavir, and darunavir are also in clinical trials for COVID-19.12,13 The combination of lopinavir and ritonavir was also in clinical trials but seems to be ineffective against SARS-CoV-2 and the use of lopinavir/ritonavir shows hardly any effect on mortality rates for hospitalized COVID-19 patients.17,18 Darunavir (a protease inhibitor) currently in phase 3 clinical trial in combination with cobicistat (a pharmacokinetic enhancer) increase the activity of darunavir by inhibiting CYP3A4).13 However, a few studies have reported that darunavir does not show any activity against SARS-CoV-2 in vitro but it is too soon to comment on the role of darunavir in patients.19 The other NRTIs (abacavir, lamivudine) and the PI (atazanavir) may target the RdRp and 3C-like proteinases in SARS-CoV-2 respectively but these drugs currently are not in clinical trials to our knowledge. Other classes of compounds like NNRTIs (efavirenz), INSTIs (bictegravir, elvitegravir, and dolutegravir) are not involved in any clinical phase trials and their effect on SARS-CoV-2 is not yet established to our knowledge.
Given the significance of the high number of people living with HIV globally, here we discuss available data on co-infection of HIV and SARS-CoV-2, their incidence, fatality rates, and the possible effectiveness of antiretroviral therapy (ART) against COVID-19.
METHODS
Information sources and literature search
A detailed literature search was performed, and the reports were taken into consideration from 1st December 2019 to 28th February 2021. A comprehensive literature search from PubMed, preprint databases (bioRxiv, medRxiv, and Preprints), reference lists, and ClinicalTrials.gov was done to identify articles for inclusion. The keywords/subject headings used for the relevant literature were “HIV (Human immunodeficiency virus) AND COVID-19 (Coronavirus)”, OR HIV and SARS-CoV-2. Descriptive analysis was conducted as per the ‘rapid review’ methodology.20 The rapid review simplify the components of the systematic review process to produce information in a short period of time.20
Inclusion and exclusion criteria
Case studies, case series, cross-sectional studies, cohorts, editorials, and short letters that reported single or aggregate clinical data on the coinfection of HIV and SARS-CoV-2 were considered. Articles (reviews, newspaper articles) not reporting original clinical data on the HIV and SARS-CoV-2 coinfections were not included in the current study. Articles in the English language were considered while those in other languages were excluded.
Data extraction
The data was extracted, reviewed, and validated by RC. Data were extracted based on publication date, study type, country, gender, and sample size. The severity index for COVID-19 for patients was classified as asymptomatic (if specified in the articles), mild/moderate (either specified in the articles or if the patient did not require admission to ICU/supplemental oxygen therapy), and severe/critical (either specified in the articles or if the patients were admitted to ICU/required supplemental oxygen/ventilation/died). The case fatality rate percentage was calculated by using the formula = total number of deaths among HIV and SARS-CoV-2 cases/ total number of HIV and SARS-CoV-2 cases from respective country *100.
Patient and public Involvement
Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
RESULTS
The selection of studies is shown as a flowchart in Figure 2. Our search strategy identified 1341 potential publications on HIV/SARS-CoV-2 co-infections. After screening the abstracts and removing irrelevant articles, we assessed 92 publications that met our inclusion criteria. The details of case studies, case series, cross-sectional studies, cohorts, editorials, and short letters that reported single or aggregated clinical data are shown in Table 1. From our review, the first case report of HIV and SARS-CoV-2 co-infection emerged from Wuhan (China) in January 2020 of a male in his 60s suffering from COVID-19 - related pneumonia who was eventually discharged in a stable condition.21 Another case from Wuhan with a preexisting dysfunctional immune system due to the co-infection of HIV-1 and HCV (Hepatitis C Virus) was reported to have COVID-19.22 Interestingly, unlike the previous case, this patient repeatedly tested negative for SARS-CoV-2 RNA, and later tested positive for IgM antibody against SARS-CoV-2.22 Two possible explanations for the undetectable SARS-CoV-2 RNA leading to negative RT-PCR test results were postulated by the authors; first - the patient was already on antiretroviral treatment (ART) (lamivudine, tenofovir, and efavirenz) which was considered to be effective against SARS-CoV-2; and second – the high levels of type I interferon (IFN-I) in HIV-1 may have cleared the SARS-CoV-2 infection.22 Few studies have also pointed out that SARS-COV-2 in its early infection phase seems to be more sensitive towards IFNs.23 After China, Singapore was the second country to report HIV and SARS-CoV-2 case in which the patient had mild symptoms of COVID-19 and was already on ART for HIV.24
Global distribution of HIV/ SARS-CoV-2 co-infections and case fatality rates
Currently, numerous countries are reporting HIV/SARS-CoV-2 co-infections across the globe (Table 1). A total of 39,461 cases of co-infections of HIV and SARS-CoV-2 have been reported worldwide out of which 24,452 (~62.0%) were males and 14,987 (~38%) were females. Out of the 39,461 cases, patient severity data on 3,213 (8.14%) were available amongst whom 43.8% cases (n=1409) were severely or critically ill (Figure 2). The severity of the coinfected individuals for their respective countries has been depicted in figure 3a. Two population-based cohort studies from the UK and South Africa reported the largest number of HIV/SARS-CoV-2 coinfections of 27,480 and 3,978 patients respectively (Figure 3, Plate A).5,11 Single case studies have been reported from Austria, Argentina, Cyprus, South Korea, Morocco, Peru and Uganda where the HIV patients had mild and uncomplicated case of COVID-19 with 100% survival rate (Table 1). An exception to this pattern was observed in Libya where a woman in her 80s on zidovudine died due to acute kidney injury and multi-organ failure, possibly due to COVID-19.25 Interestingly, all the patients from Argentina, Austria, Cyrus, Japan, Singapore (2 cases) and Uganda were on tenofovir, lamivudine, emtricitabine, and efavirenz therapy (Table 1). From the USA, 28 studies have reported a total of 6712 patients with co-infections of which ~13% were severely afflicted (Table 1; Figure 3, Plate A). Italy and Spain have 321 and 408 confirmed cases of co-infections with 23% and ~45% of patients having severe/critical symptoms respectively (Figure 3, Plate A). Compared to the three leading countries for HIV and SARS-CoV-2 co-infections, China has reported 138 patients for HIV/SARS-CoV-2 co-infections of which ~33% were severely/critically ill (Figure 1; Figure 3, Plate A). Other countries like France, Germany, India, and Turkey have fewer than 45 co-infection cases with the majority of them either mildly symptomatic or asymptomatic for COVID-19 (Table 1, Figure 3, Plate A).
Of the 39,461 reported cases of HIV/SARS-CoV-2 concomitant infections, 952 patients (~2.41%) died. From the total of severe/critical-coinfected cases (n=1409) 952 patients (67.6%) died. It is to be noted that we considered the deceased patients in the severe/critical category for reports that did not specify the severity status of the coinfected patients. Therefore, the exact proportion of severely/critically ill patients who succumbed owing to HIV/SARS-CoV-2 coinfection cannot be validated. We analyzed deaths amongst the co-infected patients, and the highest have been reported from the USA (n=710) giving the fatality rate of ~11%, mostly attributable to several comorbidities (Figure 3, Plate B).26–45 The second highest number of deaths were reported in South Africa (n=122) but with an overall case fatality rate of only ~3% (Figure 3 Plate B).5,42,43 A large portion of deceased patients from South Africa also had other co-morbidities like hypertension, diabetes, chronic kidney disease, chronic respiratory disease, and tuberculosis.5,42,43 Another recent study showed that a large proportion of COVID-19 deceased individuals from nine provinces of South Africa were people living with HIV (13.9% of 3088 total deaths; data not included in the graphs).46 Another report from New York with 2988 and 2410 cases of HIV/SARS-CoV-2 coinfection showed 7% and ~13% of fatality rate respectively which was much higher than for people with HIV but negative for SARS-CoV-2.43,46 Similar results were reported from a UK population-based cohort wherein HIV/SARS-CoV-2 coinfected individuals were at a higher risk of fatality than those without HIV.47 Interestingly, the study of HIV/SARS-CoV-2 co-infection from India (Pune and Tamil Nadu) showed that all seven patients had mild symptoms of COVID-19 with no reported deaths.48,49 In countries where single case reports have been reported, no deaths were recorded as all patients had mild symptoms of COVID-19, except in Libya where the reported positive patient died due to multi-organ failure (Figure 2).25
Potential use of anti-HIV compounds against SARS-CoV-2
One of the common multiclass antiretroviral therapy administered in combination (NRTIs and NNRTIs) for HIV patients is tenofovir, lamivudine, and efavirenz. From the analysis presented here, it is clear that a large number of patients with co-infections were on NRTIs (especially tenofovir) (Table 1). Our analysis indicates that a total of 697 people living with HIV were already on tenofovir combination therapies, out of which 39 patients died due to COVID-19 (Online Supplementary Document, Table S2). This suggests that people living with HIV have a survival rate of 94.4% on the tenofovir-based therapy. Another study has suggested the possibly reassuring role of tenofovir-based therapy for pregnant females in the context of COVID-19 pandemic,50 but more research is needed in this direction.
DISCUSSION
Our work here consolidates data on treatment and outcomes of individuals co-infected with HIV and SARS-CoV-2. As per our knowledge, this is the first study analyzing a large number of HIV/SARS-CoV-2 coinfected patients with a focus on their treatment regime and related outcomes. We analyzed available data from 92 publications with total of 39,461 HIV/SARS-CoV-2 coinfection cases reported worldwide. Multiple reports across the world show that the HIV patients co-infected with COVID-19 have mild symptoms (~64%) and the majority of them survive.51 The proportion of deceased HIV/SARS-COV-2 coinfected individuals from our study appears to be around 6% of total co-infected cases. Further, almost all the deceased patients from various studies had several comorbidities already known to aggravate COVID-19 severity.5,24,26,47,48 The most common morbidity in coinfected individuals was hypertension (n=7258; 18.4%) and diabetes (n=2946; 7.5%) out of 39,461 cases. For chronic respiratory diseases in HIV/SARS-CoV-2 coinfected individuals, we found that only 1901 (4.8%) suffered from it out of 39,461 patients.
Recent studies from the USA, UK, and South Africa have reported a large cohort of patients living with HIV/SARS-CoV-2 coinfections, with higher fatality rates when compared to HIV negative individuals.5,43,47,52 South Africa also has the most HIV cases (7.5 million) as reported in 2018 so the coinfection of HIV and SARS-CoV-2 is expected.(Table 1) 53 A South African study suggested that people with HIV on tenofovir had a lower risk of COVID-19 related deaths as compared to other antiretrovirals.5 However, reports from USA and UK have not shown any correlation with tenofovir as the data for ART is not available.43,47,52 Several other studies have also shown lower risks of patients developing severe symptoms and having higher survival rate on tenofovir against SARS-CoV-2 (Figure 1). On the contrary, a report from New York observed a high mortality rate of 78% in HIV/SARS-CoV-2 co-infection, although the sample size was very low (9 patients).27 Most of the patients with HIV are currently on ART and several studies claim that tenofovir could improve the symptoms of COVID-19.54,55
A recent pilot, randomized trial has shown promising results of the tenofovir-based therapy in decreasing the SARS-CoV-2 viral burden in recently infected COVID-19 outpatients.56 This tenofovir-based combination is reassuring including during pregnancy in COVID-19 patients.50 Tenofovir-based therapies are already in phase 3 clinical trials for prevention (NCT04334928) and treatment (NCT04712357; NCT04359095; NCT04890626) and possibly might provide an alternative to the current anti-COVID therapies for better survival rates.11,57 However, it is reasonable to suggest, as by Mirzaei et al, that continued use of ART in people living with HIV does not lead to lower morbidity and mortality from COVID-19 and several studies/clinical trials point out that NRTIs, NNRTIs, and PI classes of drugs have not shown any significant difference in morbidity and mortality patients with and without COVID-19.58,59 The impact of the tenofovir-based therapies on HIV and SARS CoV-2 requires a more systematic enquiry and longitudinal follow up either in the form of prospective cohort studies or well planned and executed randomized controlled trials. The evidence is exploratory and provides insight into the possible benefit that can be hypothesized and considered for better-designed studies. Also, the survival benefit of the regimen will need to be presented and adjusted for several factors taking into account disease severity, concomitant comorbidities, demographic factors, compliance to treatment regimens, and access to care, which also varies by setting.
COVID-19 related deaths are associated with immunocompromised status and are also dependent on several factors like age, comorbidities, healthcare facilities, etc. In the current study, the proportion of patients with HIV appear to have a high incidence of multi-morbidities, severe disease, and a higher proportion of death. We have discussed that a large number of the deceased coinfected HIV/SARS-CoV-2 patients had multiple comorbidities apart from HIV. The large number of people with HIV succumbed to SARS-CoV-2 infections having multiple comorbidities displays that COVID-19 related deaths in people with HIV might also be associated with the current immune status, age, comorbidities, healthcare management, location/country (with better healthcare facilities), and severity of the disease. However, the available data from other countries (India, Nigeria, Uganda, and Brazil) with a high burden of HIV do not indicate that the disease course of COVID-19 differs in people living with HIV than those without HIV.60
The current study has a few limitations: 1) some of the countries with a high burden of HIV (India, Nigeria, Uganda, and Brazil) reported only a few studies of co-infections with SARS-CoV-2; 2) the sample size varies across the globe; 3) the healthcare spending and systems of various countries can affect the outcomes of patients; 4) the heterogeneity among the global population is another factor that creates a bias for directly comparing such diverse countries; 5) the variables from the case reports/case series did not provide multiple factors (like RT-PCR validation of the COVID-19 reports, their cytokine profiles, alterations in the CD4+ T cells levels, etc.) and cannot be considered fully reliable. It was not possible to analyze all the variables that were of interest since not all studies mentioned the outcome, severity, and treatment regimen. The evidence generated from these case reports/series or other descriptive studies is weak and aids in generating hypothesis that should be tested by well-designed studies. This can be done through cohort studies or randomized controlled trials with the inclusion of comparison groups. Further, the role of ART on the outcome of HIV/SARS-CoV-2 coinfection has also not been fully investigated. The authors have not factored in these possible biases.
We previously proposed web-based dashboards like PRACRITI that can be utilized for predicting effective reproduction number (Rt) of SARS-CoV-2.61 Similar tools can be used to predict the SARS-CoV-2 transmission in the vulnerable group like people living with HIV to analyze the underlying effects of coinfection in the infected individuals. It is to be noted that HIV/SARS-CoV-2 coinfected patients should continuously monitor their oxygen levels with pulse oximetry for respiratory disease management. The early detection of low oxygen levels can also forewarn patients for ‘silent hypoxia’ as well and give an indication of deteriorating conditions of HIV/SARS-CoV-2 coinfected individuals.62 It has been suggested that the levels of vitamins like vitamin D correlate with the outcome of COVID-19 patients 63 while HIV-infected individuals also are vulnerable to low levels of vitamin D.64 This further highlights the vulnerability of people with HIV that can be coinfected with SARS-CoV-2.
CONCLUSIONS
As per our analysis, it is evident that HIV/SARS-CoV-2 coinfected patients have a higher risk of severity and mortality when compared to those without HIV and other comorbidities as is evident by the large cohort studies. The spread of both viruses coexisting in the vulnerable populace might lead to adverse public health outcomes. Further, people with HIV on a tenofovir regimen if coinfected with SARS-CoV-2 might have a better survival rate than those on other ART regimens. It is a possibility that needs to be tested further that people with HIV already on the antiretroviral regimen (especially tenofovir-based therapy) may display some level of co-protection against SARS-CoV-2 infections and the individuals might develop mild symptoms. In sum, HIV/SARS-CoV-2 co-infection cohorts need to be analyzed across many countries with high HIV burdens with much larger data sets.
Data Availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Patient and Public Involvement
Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
Funding
None
Author contributions
RC collated, analyzed and interpreted the data; drafted the manuscript. SM analyzed and edited the manuscript critically. AS conceptually designed the work, interpreted the data, drafted the manuscript and revised it critically.
Competing interests
The authors completed the Unified Competing Interest form at http://www.icmje.org/disclosure-of-interest/ (available upon request from the corresponding author), and declare no conflicts of interest.
Correspondence to:
Amit Sharma, Molecular Medicine Group, International center for Genetic Engineering and Biotechnology, New Delhi, India.