Global measures to reduce premature mortality have contributed to a rise in life expectancy: people live longer and a growing proportion live with more than one chronic condition, a phenomenon commonly referred to as multimorbidity.1 Previous research has found that individuals with multiple chronic conditions have complex healthcare needs and are characterised by deteriorated physical, social and mental wellbeing, higher premature mortality, poor quality of life and increased utilisation of healthcare and social services.2,3 One of the largest prevalence studies established that multimorbidity is associated with low socio-economic status and that individuals from deprived areas were likely to develop multiple chronic conditions ten to fifteen years earlier than those from more affluent settings.4
Although most evidence comes from high income countries (HICs),5 it is evident that the burden of multimorbidity is rising and putting pressure on health care systems and economies worldwide.6 Prevalence estimates range from below 15% to above 95%.7 A recent systematic review summarising global literature on the prevalence of multimorbidity based on seventy community based studies, estimated a prevalence of 37.9% for HICs and 29.7% in low- and middle-income countries (LMICs).8 Understanding and mapping the magnitude of multimorbidity, as well as how diseases co-exist is critical in allocation of resources in healthcare and development of effective, efficient evidence-based management and prevention strategies.8,9 The literature has pointed to the lack of international consensus on the operational definition of multimorbidity as well as the selection of conditions that meet the chronicity criteria as the major reason behind varying results that are difficult to compare across different settings.1,7,10
Multimorbidity in the sub-Saharan Africa context
Although many countries especially HICs have managed to significantly reduce the prevalence of communicable diseases, in the sub-Saharan Africa region there remains a significant burden from diseases such as tuberculosis (TB), human immunodeficiency virus (HIV), malaria and these remain the leading causes of mortality and morbidity.11,12 In parallel, the prevalence of NCDs in SSA is rising - primarily cardiovascular diseases, chronic respiratory disease, diabetes, mental disorders and cancers. Due to rapid urbanisation, adoption of risky lifestyles such as harmful use of tobacco and alcohol, lack of exercise and poor diet, the region has experienced a rise of approximately 67% in the burden of NCDs between 1990 and 2017.13 Furthermore, due to the success of antiretroviral treatment (ART), survival in people living with HIV has significantly improved14 and research shows that these individuals have an increased risk of developing chronic conditions such as cardiovascular diseases, depression, cancers among others.15,16
This increased prevalence of NCDs among people living with HIV has been linked to various factors including the nature of HIV infection, effects of certain antiretroviral drugs, ageing and other well-known NCD risk factors.17 This interaction presents a complex form of multimorbidity that is different from the one observed in HICs. For example, SSA is likely to have a larger proportion of an ageing population on ART in the coming decades.18 This highlights the importance of generating context-specific evidence for SSA, a region with the highest global burden of HIV infection which is, concurrently, experiencing a rapid increase in the burden of NCDs.
Rationale
Although there is clear evidence that the prevalence of multiple chronic conditions is rising in all regions of the world including sub-Saharan Africa (SSA), there is still limited evidence from the region to give a clear picture of the status or burden of multimorbidity. Most systematic reviews on prevalence and patterns of multimorbidity to date have focused on HICs and those that included SSA countries grouped them together with other countries in the ‘LMIC’ category. Evidence is needed specifically for this region due to the unique disease profile of a double burden of communicable diseases and NCDs. Given the rapid population growth in the region, an ageing HIV-positive population, the interaction of NCDs and communicable diseases, and already existing health care challenges, it is imperative to generate SSA-specific evidence.
The overall aim of this review is to identify and synthesise the evidence on the prevalence and patterns of chronic disease multimorbidity among adults in sub Saharan Africa by examining the interaction of chronic communicable diseases such as HIV, TB with the most prevalent NCDs in SSA. We will also evaluate the evidence on how multimorbidity in the region is typically measured, and how the differences in measurement might influence reported patterns of multimorbidity. With the advent of the COVID-19 pandemic, we will also incorporate emerging literature on the impact of this pandemic on chronic conditions. To the best of our knowledge, this is the first systematic review that will map evidence on the combined burden of non-communicable and communicable disease multimorbidity specifically for SSA region.
METHODS
The development and reporting of this protocol was in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analyses Protocols (PRISMA-P) 2015 statement.19 This protocol has been published with the International Prospective Register for Systematic Reviews (PROSPERO), registration number CRD42020183539.
Eligibility criteria
The inclusion criteria is guided by the CoCoPop (condition, context and population) framework for reviews of prevalence studies recommended by the Joanna Briggs Institute.20,21
Condition: We will adopt the Academy of Medical Sciences definition of multimorbidity: the coexistence of two or more chronic conditions, each one of which is either; a physical non-communicable disease of long duration, such as a cardiovascular disease or cancer, a mental health condition of long duration, such as a mood disorder or dementia, or an infectious disease of long duration, such as HIV or hepatitis C.22
Context: We will consider studies from the sub-Saharan Africa region. Sub-Saharan Africa will be defined according to the World Bank country classification23 as illustrated in Table 1.
Population: We will include multimorbidity studies involving adults aged 18 years and older.
Studies will be selected based on the following inclusion criteria;
-
Original studies aimed at estimating the prevalence of multimorbidity or documenting the frequent disease patterns conducted either at community or health facility level.
-
Observational studies including cross sectional and longitudinal studies. For longitudinal studies we will extract prevalence at baseline.
-
Government and non-governmental surveillance data/reports and theses that report original data on prevalence or patterns of multimorbidity.
Due to the paucity of research on multimorbidity in sub-Saharan Africa, studies that selected participants based on a specific condition (comorbidity studies) will not be excluded since it is important to understand how the two concepts have been defined and measured in the region. Distinctions on the characteristics of comorbidity and multimorbidity studies will be reported accordingly.
We will collect data from studies published between January 2000 through to December 2020. The rationale behind this date range selection is that global research on multimorbidity only started increasing in the early 2000s, with 80% of studies published after 2010.5,24 We therefore anticipate that before 2000 there were very few originating from sub-Saharan Africa, especially considering that even after 2010 only approximately 5% of research in this field comes from LMICs.24 Furthermore, the availability of ART after 2000 changed the HIV situation in the region, making it a manageable chronic condition,25 and considering the scope of research around HIV comorbidities and NCDs we deem this date range most appropriate for the sub-Saharan Africa context. We will apply no language limits on our literature search; studies in other languages will be translated to English.
We will exclude studies conducted among people below the age of 18 years and those from countries outside the sub-Saharan Africa region. Articles not reporting original peer-reviewed research such as abstracts, conference presentations, opinion reports and reviews will also not be included.
Search strategy
To identify appropriate and relevant studies for the review, the following databases will be searched; MEDLINE, Embase, Cumulative Index of Nursing and Allied Health Literature (CINAHL), Global Health, PsycINFO, African Journals Online and African Index Medicus. We will also hand search reference lists of relevant studies to identify further literature of interest. Additionally, we will use ProQuest Dissertation & Theses, Web of Science, Google Scholar and Google search engine to identify grey literature such as government and institutional reports, theses and dissertations, as well as to track citations. Key authors for studies that meet eligibility criteria will be contacted if there is some missing information or full texts of their studies cannot be accessed.
A comprehensive search strategy and terms was developed by the team in collaboration with the University of Edinburgh Medical Librarian. We will conduct a broad search, adopting some search terms from previous systematic reviews on the topic.7,8,26 Search terms will include “multimorbidity”, “comorbidity”, “prevalence”, “sub Saharan Africa” and their synonyms. Medical subject headings (MeSH) and key texts words will be developed and combined with Boolean operators “AND” and/ “OR” across and within categories. A full search strategy for MEDLINE (Ovid) database is provided in Table 2, and will be tested and adapted to other databases.
Data collection and analysis
Selection process
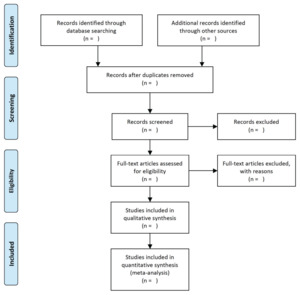
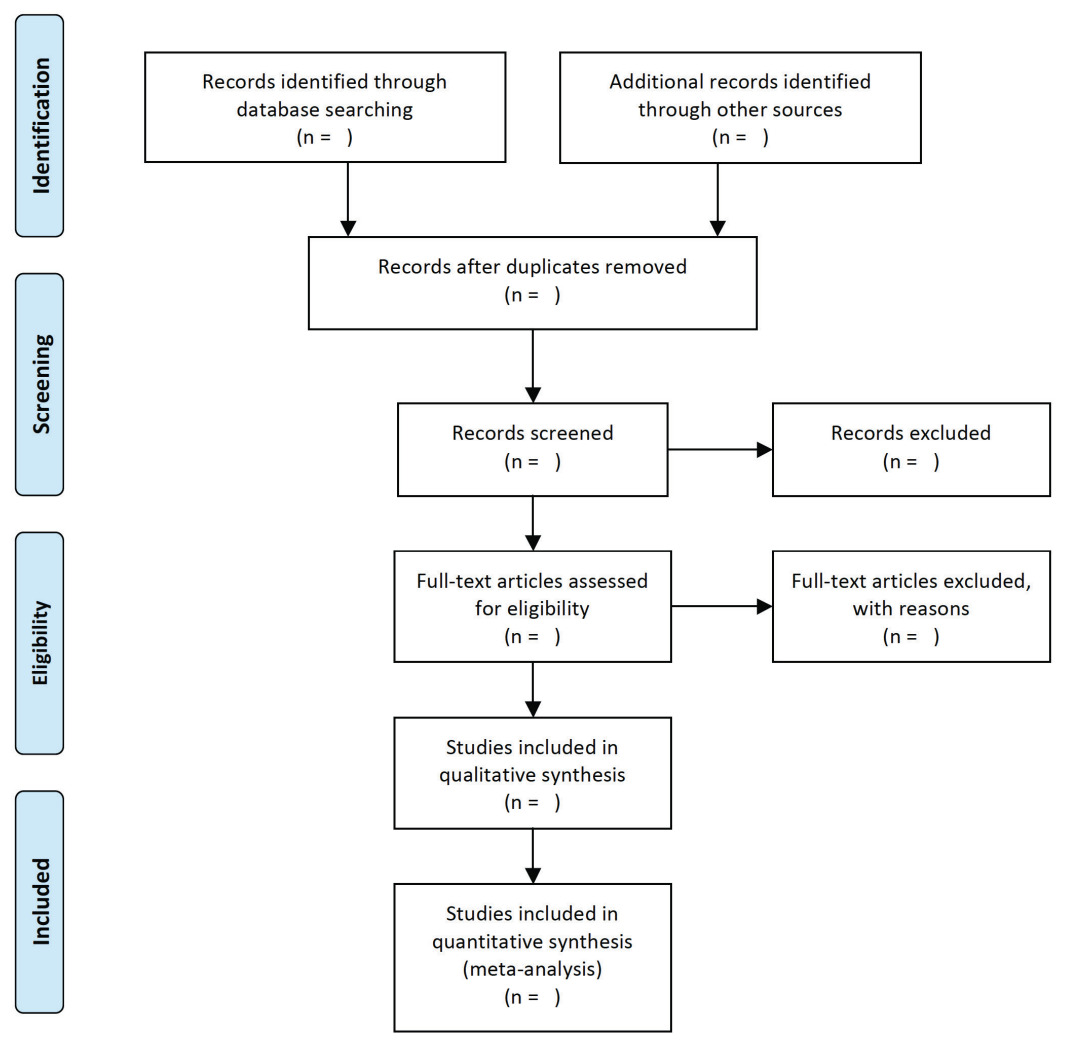
For the first stage of the review, titles and abstracts of potentially eligible studies identified through electronic database searches, will be extracted to Endnote Library (EndnoteX9). Duplicates will be removed (both via Endnote function, and manual checking). Two reviewers will then independently review the titles and abstracts of the remaining studies to identify studies that potentially meet the inclusion criteria. If any discrepancies arise the two reviewers will discuss them and reach an agreement, with a third reviewer providing input. Selection of full text studies against the eligibility criteria will be carried out independently by the two reviewers. The third reviewer will also randomly check 10% of selected studies to check for consistency. Reasons for excluding studies will be documented throughout the process. A PRISMA flow diagram (Figure 1)27 will be used to outline the literature search and selection process.
Data extraction
Data extraction from selected studies will be carried out independently by two reviewers. In the event that there are disagreements, there will be discussions between the two reviewers and if a consensus is not reached the third reviewer will adjudicate. The team will develop a data extraction form which will be pre-tested. Key study characteristics are shown in Box 1.
Quality assessment
We will evaluate the overall quality of individual studies using the Joanna Briggs Institute Prevalence Critical Appraisal Tool.28 This tool appraises external and internal validity of each individual study by addressing issues of representativeness of sample, recruitment of participants, identification of the condition, its measurement and statistical analysis among others. Two reviewers will independently assess the quality of included studies and disagreements will be resolved through discussions with a third reviewer.
Data analysis
We will conduct a narrative synthesis of data from all included studies using “Guidance On The Conduct of Narrative Synthesis In Systematic Reviews”.29 Studies will be clustered according to multimorbidity definition (2 or more chronic diseases, or 3 or more chronic diseases), setting (population based and facility based). We will generate evidence tables to summarise, descriptively, study and participants’ characteristics. Characteristics of comorbidity studies versus multimorbidity will also be captured in table format. We anticipate that potential studies that will meet the inclusion criteria, are likely to be heterogeneous in different features such as study population, settings and data sources hence we expect that meta-analysis might not be suitable.30 However, if there are sufficient data that we are able to synthesise through meta-analysis, we will estimate a pooled prevalence of multimorbidity using random effects model together with their 95% confidence intervals (CI). Heterogeneity of studies will be assessed using the I2 statistic. An I2 value of 25%, 50% and 75% indicating low, medium and high heterogeneity, respectively.31 For any data with substantial heterogeneity, we will consider sub group analysis using mean age, sex, study setting, World Health Organisation Region in Africa, definition of multimorbidity.
Ethical considerations
This study does not require ethical approval as only secondary data will be assessed and does not involve interaction with the study subjects.
CONCLUSION
This review will identify and summarise evidence on the burden of multimorbidity in SSA taking into account the context of the region which is characterised by a double burden of communicable and non-communicable diseases. Results of this review will be published in peer reviewed journals and shared through relevant academic conferences.
Acknowledgements
We thank Marshall Dozier (Academic Support Librarian (Medicine), at the University of Edinburgh) for helpful discussion on search strategy design.
Funding
This is the first component of a PhD project which is examining the magnitude of multimorbidity and the common disease combinations in SSA.MRD is a PhD student at the Usher Institute, University of Edinburgh and is funded by the University of Botswana (UB). UB had no role in preparation, funding or decision to publish this protocol paper.
Authorship contributions
MRD made substantial contribution to the conception of the protocol, drafting the manuscript and incorporated comments and suggestions from co-authors. DW and CC revised the drafts critically for important intellectual content and approved the final version for submission.
Competing interests
The authors completed the Unified Competing Interest form at www.icmje.org/coi_disclosure.pdf (available upon request from the corresponding author), and declare no conflicts of interest.
Correspondence to:
Mpho Refilwe Disang
Usher Institute
University of Edinburgh
Old Medical School
Teviot Place
Edinburgh, EH8 9AG
United Kingdom