Low- and middle-income countries (LMICs) contribute over a half of global cancer burden in terms of both incidence and mortality.1 The increase of cancer rates between 2008 and 2030 in LMICs is expected to be greater than in high income countries (76% vs. 25%).2 Cancer survival rates in LMICs vary according to health care access and service delivery3; for example, 5-year rates are higher in China and Turkey, which have better healthcare availability and access, compared to countries such as India or Thailand.3 Survival rates also vary according to the site, for example, low survival is observed for cancers of the stomach, lung and tongue.3
Cancers in India represent over 10% of the total LMIC burden, with an estimated 1,157,294 incident cases and 784,821 cancer deaths reported in 2018.4 Recent cancer site-specific burden assessment revealed substantial heterogeneity in the state-level incidence rates and health loss trends over a 26-year period (1990-2016).5 It is also notable that most cancer deaths in India are reported to occur at home without access to medical care.6 This complicates tracking of cancer survival, which is essential for cancer control efforts, and yet too resource-intensive for many cancer registries in India.
Survival data may reflect timing of detection, stage at diagnosis, and treatment outcomes; thus helpful in making context-specific decisions for efficient and cost-effective cancer care.3,7 Despite the increasing cancer incidence and mortality,8 survival data in India are limited to a few population-based cancer registries located in the Southern and Central parts of the country9–26 and the available cancer outcomes research is limited to a few hospital-based studies.27–31 Sparse data are available on cancer survival in Northern India, particularly for primary sites with very poor prognosis.
The existing knowledge gaps on cancer survival in LMIC generally, and India in particular, and the difficulty of collecting prognostic information through cancer registries, motivated development of complementary methods for patient follow-up in a resource-intensive setting.3,6 Lung (LC) and gallbladder (GBC) cancers are highly lethal cancer sites commonly affecting men and women in Northern India.8 While LC is a leading cancer site among men paralleling global pattern, GBC is a very rare digestive tract cancer in other parts of the world.8 High GBC incidence is uniquely common in this region especially among women.8,32 In the present study we evaluated the feasibility of using telephone surveys as a method of active follow-up for collecting survival data for these leading and lethal cancer sites in Northern India. We also use this methodology to estimate absolute survival rates for LC and GBC.
METHODS
This is an ambidirectional cohort study of LC and GBC patients registered at the Postgraduate Institute of Medical Education and Research (PGIMER). PGIMER is a premier government tertiary care hospital in Chandigarh, a Northern Indian union territory. PGIMER established a hospital-based cancer registry (HBCR) in 2011.
The sampling frame for the study included all patients reported to the HBCR who were diagnosed and/or treated between 2011 and 2014 and had a valid telephone contact, and stratified random sampling was used to select the study participants within two 2-year intervals 2011-2012 and 2013-2014. Patients who were diagnosed and/or treated in 2010 were selected from PGIMER medical records.
Male and female cancer participants were considered eligible for inclusion in the study if they were 30 to 69 years of age at the time of diagnosis or registration in PGIMER and had primary malignancy of the lung (C34) or gallbladder (C23). Participants were excluded if they did not have at least one valid telephone number (landline/mobile) available in the records. The eligible patients resided primarily in Chandigarh, and in the nearby states of Punjab, Haryana, and Himachal Pradesh, although some also came from other areas such as Bihar, Uttar Pradesh, Uttarakhand and Jammu and Kashmir.
The resulting study population included 530 eligible patients (259 LC and 271 GBC). Information on sociodemographic factors (age, sex), contact information (address and valid telephone number), diagnosis (stage, clinical extent before treatment, method of confirmation, and histology), treatment receipt (surgery, radiotherapy, chemotherapy, combined, palliative or other) and follow-up was abstracted from medical records.
Participants (or their family/relatives) were followed-up with telephone interviews between 01-03-2016 and 31-07-2016 and the vital status of cancer patients was recorded. Surviving participants or their care takers were interviewed directly and for participants who were not alive at the time of interview, the date of death was recorded from family or close relatives.
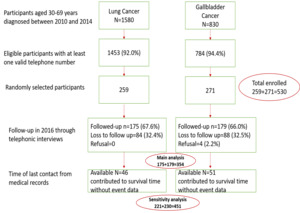
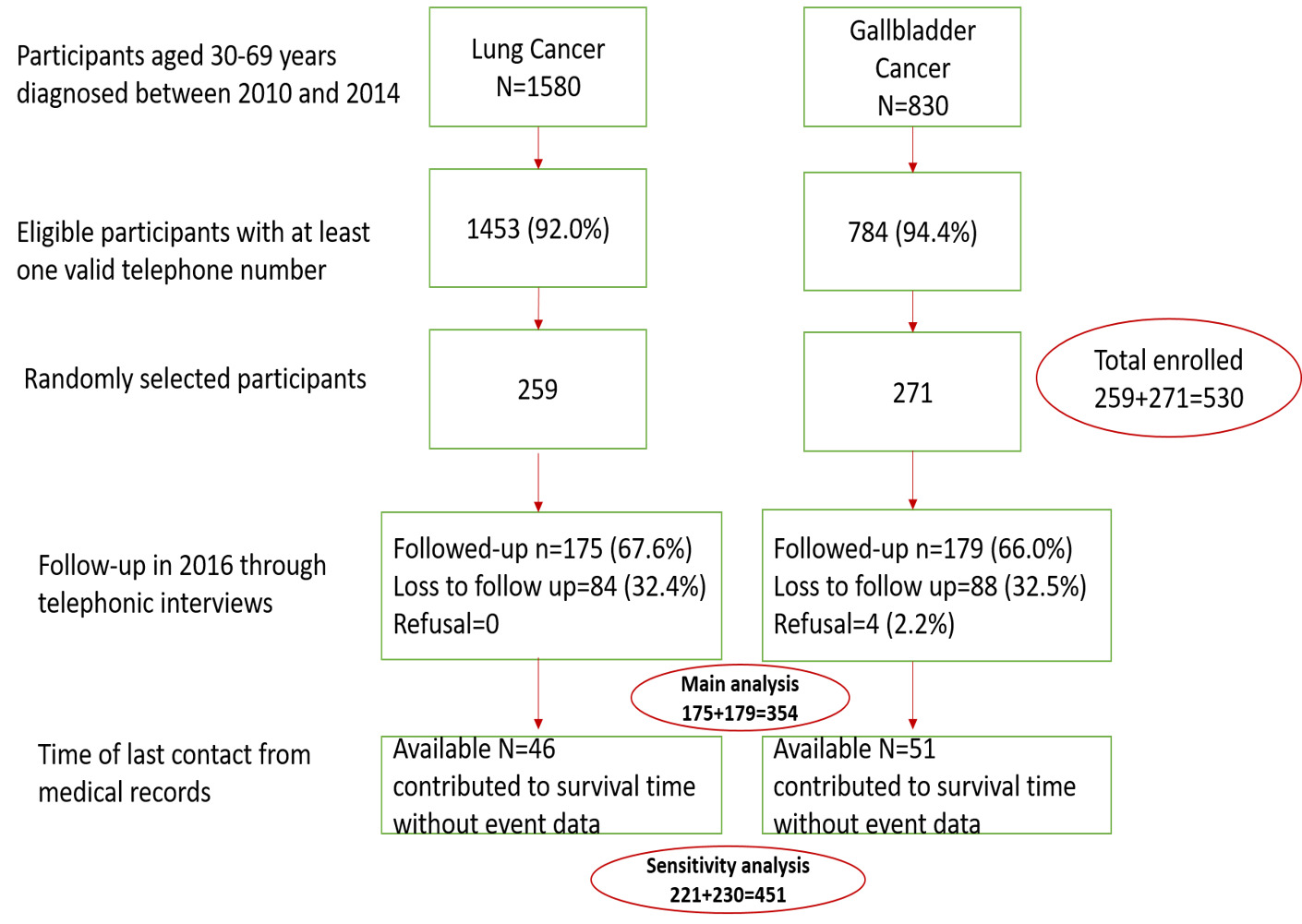
Out of 530 eligible patients, 358 participants or their families were contacted and 354 agreed to participate in the telephone interview. Out of 176 who were lost to follow-up (33.2%), 97 participants contributed to survival time without event data based on last follow-up information in hospital records. Figure 1 summarizes the information of eligibility, recruitment, participation and losses to follow-up.
As this was a relatively novel approach to collecting survival data, a record of implementation logistics was maintained to evaluate the feasibility of using telephone calls for cancer surveillance in India. The various elements of study implementation included: number of telephone call attempts, reason for success/failure of each attempt, reason for multiple attempts, and available number of participants who answered the call by day of week, time of day, time of call, person. Reasons for not answering the calls were also documented; these included faulty phone number, patient not reachable, patients not available, or reaching someone unrelated to the patient. Each telephone number was dialled up to 5 times over a period of 2 weeks at different times of the day and different days of the week before it was declared invalid.
For the purposes of data analyses, the follow up of study participants started on the date of LC and GBC diagnosis and ended at the time of death or the time of the last interview. One- , three- and five-year survival rates were calculated using Kaplan-Meier method. Weighted Cox Proportional-Hazards regression multivariable models accounting for loss to follow-up were used to evaluate independent associations between various factors and survival with results expressed as adjusted hazard ratios (HR) and the corresponding 95% confidence intervals (CI). The independent variables of interest in these models included age at diagnosis, sex, education, clinical extent before treatment and histology (for LC only). Proportional hazards assumptions were tested using log-log curves, curves for observed-predicted values with global test and Snell’s residuals. Interactions between sociodemographic variables (age at diagnosis and sex) and clinical variables (clinical extent and histology) were tested using likelihood ratio tests. The same survival curves and Cox regression models were used in a separate analysis that include participants who were lost to follow-up but contributed to survival time without event data (Figure 1). All statistical analyses were conducted using STATA software version 10 (StataCorp Inc, College Station, TX, USA).
Ethical approval was obtained from respective institutional ethical committees TRC-IEC 268 (PHFI) and IEC 03/2015-168 (PGIMER). Informed consent was obtained from participants and their family members and relatives over the phone.
RESULTS
The baseline characteristics and follow-up status of the eligible patients (N=530) are summarized in Table 1. Average age at diagnosis (standard deviation) was 53.3 (±8.9) years; 58.1% were men; and 37.9% had no formal education. About half had an advanced pre-treatment clinical extent (48.7%); and only 57.3% underwent cancer directed treatment. Most LC participants were men (85.4%) while approximately two-thirds of GBC participants (68.3%) were women. In comparison with LC participants, GBC participants were on average younger when diagnosed (51.4 vs. 55.3 years), were less educated (46.5% vs. 29.0% with no formal education), and were more likely to present with advanced clinical-extent of disease (56.1 vs. 40.9%) or receive no treatment (52.8 vs. 13.5%). However, there was no difference in the active follow-up through telephone calls between the two groups of participants.
Feasibility of telephone follow-up
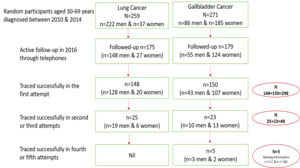
Over two-thirds of cancer patients or their families (n=354; 66.8%) were successfully interviewed over the telephone (65.9% men & 68% women) (Figure 2). An examination of call logs showed that 84.2% of participants (n=298) were contacted on the very first attempt. Another 13.5% were contacted on either the second or the third attempt (n=48) and for the remaining 5 participants (1.4%) contact was established after four or five calls (Figure 2). The follow-up patterns did not differ between men and women. In a sub-set of participants (n=260) we recorded information on the type of phone service they currently used. All of them used mobile phones; 65.8% used pre-paid mobile service and 10.8% used post-paid service, 23.5% did not know the kind of mobile service they used. For about 33% of eligible patients telephone contact was unsuccessful (n=84 LC participants & 88 GBC participants). A comparison of baseline characteristics of patients followed-up by telephone and those lost to follow-up is presented in Table 2. The two groups did not differ with respect to demographic factors, education, disease characteristics, and treatment status. The top two reasons for inability to contact the participant or their family members were: (1) the number was abandoned or reallocated to different users (57.9%) and (2) the number did not exist or not reachable or went unanswered after 5 attempts over 2-week period (37.4%).
Survival analyses
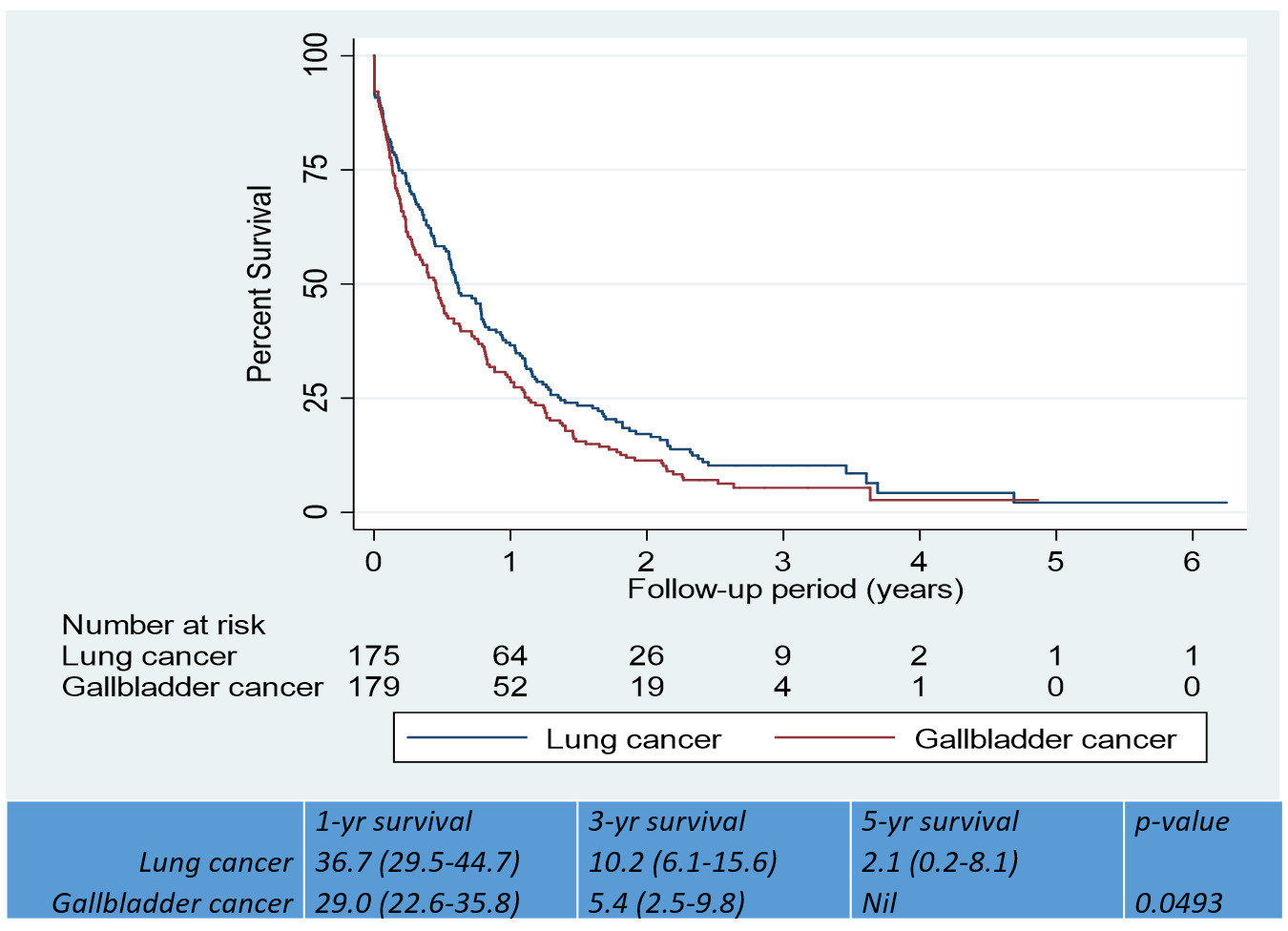
Survival analysis included 354 participants (N=175 LC; 179 GBC) who were actively followed up through telephone interviews or had a recorded date of death. The total follow-up time was 303.5 person-years. A total of 326 deaths (158 LC; 168 GBC) were observed during the observation period. Absolute 1-year, 3-year and 5-year, survival rates, expressed as percentages (95% CIs) for LC were 36.7 (29.5-44.7), 10.2 (6.1-15.6) and 2.1 (0.2-8.1) respectively. The corresponding 1–year and 3-year estimates for GBC were 29.0 (22.6-35.8) and 5.4 (2.5-9.8) with nil survival for 5-year term. GBC had lower survival rates than LC (p=0.049) (Figure 3). Absolute survival rates by baseline characteristics for each cancer site are presented in Table 3. LC participants experienced better survival in the intermediate age group (41-50 years) than in the younger (30-40 years) or the older age groups (51 years and above). GBC participants with localised clinical extent had better survival than the rest Table 3.
In weighted multivariable Cox regression analysis, advanced pre-treatment clinical extent was associated with reduced survival; using localized disease as reference, the HR (95% CI) estimates were 2.0 (1.3, 3.1) for GBC and 1.5 (1.0, 2.2) for LC (Table 4).
When we re-examined the same associations by including participants who contributed to survival time without event data (N=451), the results were consistent with those observed in the main study population. Additionally, in weighted multivariable Cox regression analysis, graduate education was associated with improved survival in LC; using no formal education as reference the HR (95% CI) estimate was 0.4 (0.2, 0.9) (Table S1 in the Online Supplementary Document). A comparison of baseline characteristics between patients who contributed to survival time and those who did not revealed no significant differences (Table S1 in the Online Supplementary Document).
DISCUSSION
Our study indicates feasibility and acceptability of telephonic approach for active surveillance of cancer survival in India. We recorded a response rate of 66.7% with few refusals (1%).
The logistics of implementing this methodology are complex and require detailed planning and co-ordination. Of particular importance is capturing accurate personal and contact information. We found this very challenging particularly for participants diagnosed and registered in 2010, because systematic data collection through HBCR was not available for that year. While over 90% of participants had a valid telephone contact number between 2011 and 2014, this information was available for less than a half of participants in 2010. The major reasons for inability to follow-up of participants who had a valid telephone contact, were disuse and reallocation of existing numbers. All participants had a mobile contact number (only one participant had 2 landline numbers and was lost to follow-up). In a sub-set of participants, we found a majority used pre-paid mobile service. As permanent mobile numbers are increasingly more common, the challenges of reallocation and disuse may become less important.
Continuous surveillance of cancer survival in many developed regions of the world is possible due to systematic data collection via both passive and active follow-up.7 Tracking cancer patients for vital status and quality of life measures through telephone interviews is widely practised in many developed33–37 and in certain developing countries.38–40 The widespread use of mobile technology, creates an opportunity to collect data on vital status with relatively few resources.41 For many registries in India and other LMICs, it may serve as a cost- and time- efficient data collection method.41 The telephone interview method may help maximize survey response and completion rates, and therefore yield population-based data of higher quality.42 Telephone contact may offer advantages over face-to-face contact, in terms of convenience, confidentiality and continuity, all of which are important for cancer patients.43
Our data indicate that both LC and GBC diagnoses are associated with poor prognosis in Northern India with less than 3% survivors for LC and no survivors for GBC at the end of 5 years. Hospital-based survival estimates may differ from population-based estimates in several ways.44 For example, the population-based 5-yr survival rates for LC were reported to range between 7%3 and 9.6%7 in Indian population-based cancer registries, compared to 2.1% in our study. This discrepancy may be attributable to higher proportion of LC cases with advanced disease reported to hospital registries; however, similar comparisons are unavailable for GBC.
In our study ‘clinical extent of the disease’ was the only discernible predictor of survival, especially for GBC. In addition, age at diagnosis and education appeared to be related to LC survival, which requires further evaluation. Demographic and socioeconomic factors, reflecting the disparities in the levels of health education and in accessibility and affordability of hospital care are important determinants of cancer survival both across India and worldwide,3,7 in addition to clinical and treatment parameters.
LC is one of the leading causes of cancer mortality worldwide both in developing and developed regions7; even in Europe, North America and Australia where advanced treatments are often available, 5-year survival rate is reported to range between 15 and 20%. In India, lung is the second most common cancer site in men4 and an emerging site in women with relative change of 136.1% reported between 1988 and 2005.45 With 85% of cases in India reported to have late diagnosis, LC mortality and associated morbidity are very high.46
GBC is a highly lethal malignancy, which is relatively rare worldwide, but common in Northern and Eastern regions of India5 across the Gangetic belt (ie, Assam, Delhi, West Bihar, East Uttar Pradesh) with women more commonly affected.5 Trends of pooled data from 5 urban cancer registries for the period (1988-2005) suggest a relative increase of 162% in GBC incidence among women.45 Despite the alarming increase in disease burden, continuous surveillance of cancer survival is very limited in India. Continuous surveillance of cancer survival will be an indispensable source of information for multiple stake-holders including cancer patients, health care providers, researchers and policy makers.
This study is not without limitations. First, eligible participants were those who had a valid telephone number. This may be of concern if survival rates among participants without a valid contact were different perhaps due to lower socioeconomic status or inadequate access to care. On the other hand, less than 10% of eligible participants in our study did not have any telephone contact and majority of them were diagnosed in 2010 when HBCR was not established. Thus missing contact information was more likely due to less systematic data collection in previous years rather than socioeconomic disparities. The second concern was loss to follow-up. The impact of loss to follow-up was likely minor as we found no differences between participants with and without complete data. In addition, the effect of loss to follow up, if any, was taken into account in the weighted analyses.
Key message
We report the feasibility of using telephones for active surveillance of cancer survival in Northern India. Surveillance for cancer survival is very limited in many resource intensive settings and the present study offers new information on possible ways to evaluate cancer survival in such settings. Active follow up of cancer patients and /or family and relatives through telephone calls is feasible and acceptable in India, which could be scaled-up nationally and could also be valuable in other LMICs. The success of this approach may depend on systematic collection of required personal and contact information at hospitals and cancer registries and reaching out to family members, particularly for cancer sites with poor survival outcomes.
CONCLUSIONS
We found that the use of telephone contacts to assess cancer survival is both feasible and acceptable in Northern India. The success of this approach may depend on systematic collection of required personal and contact information at hospitals and cancer registries. Poor survival rates of both LC and GBC underscore the importance of reaching out to family members. The present study offers new information about possible ways to evaluate cancer survival in a resource-intensive setting, which could also be valuable in other LMICs. These data can assist with continuous monitoring of cancer survival and inform future cancer control efforts.
Funding: This study was funded by PHFI-PH leader Implementation Science Programme granted to Preet K. Dhillon. Funders had no role in the design, data collection, analysis or publication of the manuscript. Krithiga Shridhar was supported by a Wellcome Trust Capacity Strengthening Strategic Award Extension phase to the Public Health Foundation of India and a consortium of UK universities (WT084754/Z/08/A) during the study and is currently supported by the ‘Research Development Grant’ from the Centre for Environmental Health at the Public Health Foundation of India.
Authors’ contributions: All authors contributed equally.
Competing interests: The authors have completed the Unified Competing Interest form at www.icmje.org/coi_disclosure.pdf (available on request from the corresponding author) and declare no competing interests.
Correspondence to:
Dr. Krithiga Shridhar, MDS, MSc
Centre for Chronic Conditions & Injuries
Public Health Foundation of India
Gurugram
Haryana
India
[email protected]