INTRODUCTION
Chronic Kidney Disease (CKD) has become a mammoth public health concern, and its surge tends to have an uninterrupted triumph.1 Approximately 10% of the world population is victim of this enormous ailment. Global Burden of Disease Study- 2015, has reported renal disease was the 12th most common cause of mortality, and globally responsible for 1.1 million deaths.1,2 Prevalence of CKD in India is estimated as 13–15.04% with stage 1, 2 and 3 as 6.62%, 5.40% and 3.02% respectively.3
Hemodialysis (HD) is a decisively essential management modality in patients with end stage renal disease (ESRD) that extends the life expectancy and enhances the quality of life (QOL).4 One of the prevalence studies among HD patients estimated more than 50% are non-adherent in some ways within the first month and upwards of 70% of clients fail to successfully control their fluid ingestion.5 Such alarming rates are a matter of concern because even irregular occasion of serious non-adherence can result in lethal complications. Worldwide numerous interventions and therapies had been developed to enhance compliance to HD regimen and QOL.5–10
Griva et al pointed out the benefits of a self management intervention (SMI) on HD patients through their randomized controlled trial (RCT). This study recruited 176 adults undergoing HD at least for 6 months and they were randomized to self management or standard care groups. In this multicentre parallel arm block RCT of a four session group SMI for HD delivered by health care professionals compared to standard care. The SMI was revealed to be effective in terms of adherence and QOL.6
Chen CC et al through their meta-analysis emphasized that QOL of CKD patients improved through nurse led disease management program. They had comprised eight studies and 1520 patients, with 766 clients allocated to the nurse-led disease management schedule. It was observed nurse-led disease management significantly magnified the QOL in terms of general health perception, symptoms, pain, energy/fatigue, staff encouragement, sleep, overall health and psychological component summary when determined 6 weeks after the commencement of intervention.5 Nursing intervention consists of the appliance of behavioral, educational, cognitive and dietary techniques that has been depicted to had positive effects on the emotional and physical health of patients with ESRD.5–10
Ahrari et al in their study on ESRD patients in Iran mentioned that non-adherence to diet and fluid control is a primary reason for treatment letdown and deprived outcomes.11 However, predictors that impede with the patients’ capability to pursue their dietary control remains unidentified. Through their study about Jordanian HD patients, Khalil et al suggested that recognition of predictors that may aggravate diet and fluid non-adherence may contribute to get favorable therapeutic outcomes via explicitly developed interventions.12,13
Beerappa and Chandrababu in their prevalence study recommended the need of a well framed RCT on fluid and diet adherence in order to develop useful interventions for evidence based practice.14 A meta-analysis by Wang et al showed that nursing interventions improved patient’s adherence to HD, and the mean effect was found to be 15% in total.10 Hence, evidence based therapeutic nursing interventions, that are cost effective and readily accessible are vital for facilitating adherence behavior.10
Dietary and fluid adherences are quintessential components in the management of ESRD patients.6 Non-adherence to diet and fluids restriction may result in increased morbidity and mortality because of the accumulation of toxic fluids and end products of metabolism in the blood.5 These literatures suggest that such existing psychosocial interventions facilitate adherence and enhance self-care management among HD patients. However majority of such studies are methodologically underpowered, intervention protocols are poorly defined and developed, very short follow up duration and marked with lack of simultaneous inculcation of both psychological and clinical outcomes.12,14 Other criticism in reviews are the lack of detailed description of the required training, inadequate theoretical or conceptual support of these intervention that might have ultimately degrade fidelity of the existing intervention in many studies.7–10
Although certain studies pointed out different psychological interventions to fill the gap in the knowledge, unfortunately a scientific conscience is lacking.5,7–10 This vivid gap in the evidences provoked the researcher to initiate a work plan to solve this unresolved dilemma. The proposed RCT will tackle the fragility of previous counterpart studies by framing a well defined methodology, recruiting sufficiently large and representative HD patients, the development of well designed, evidence based and theory driven intervention and the inclusion of validated and reliable measurement tools to ascertain adherence and health related outcomes through subjective as well as objective methods. Hence this trial will probably endow with much needed data on the efficacy of nurse led interventions with respect to adherence and other health related outcomes that can confer evidence based clinical practice and health care services.
METHODS
Objective
The study aimed to examine the efficacy of nurse-led-interventions on dialysis related diet and fluid non-adherence and morbidities
Hypothesis
-
H1: Dialysis patients participating in nurse led intervention sessions will be more adherent to dietary and fluid restrictions compared with patients not in the intervention group
-
H2: Dialysis patients receiving nurse led interventions will have less morbidities compared with patients not in the intervention group
Approach and design
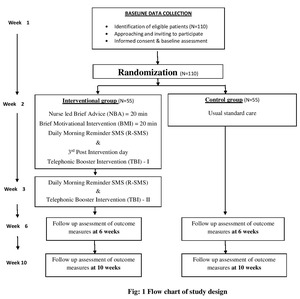
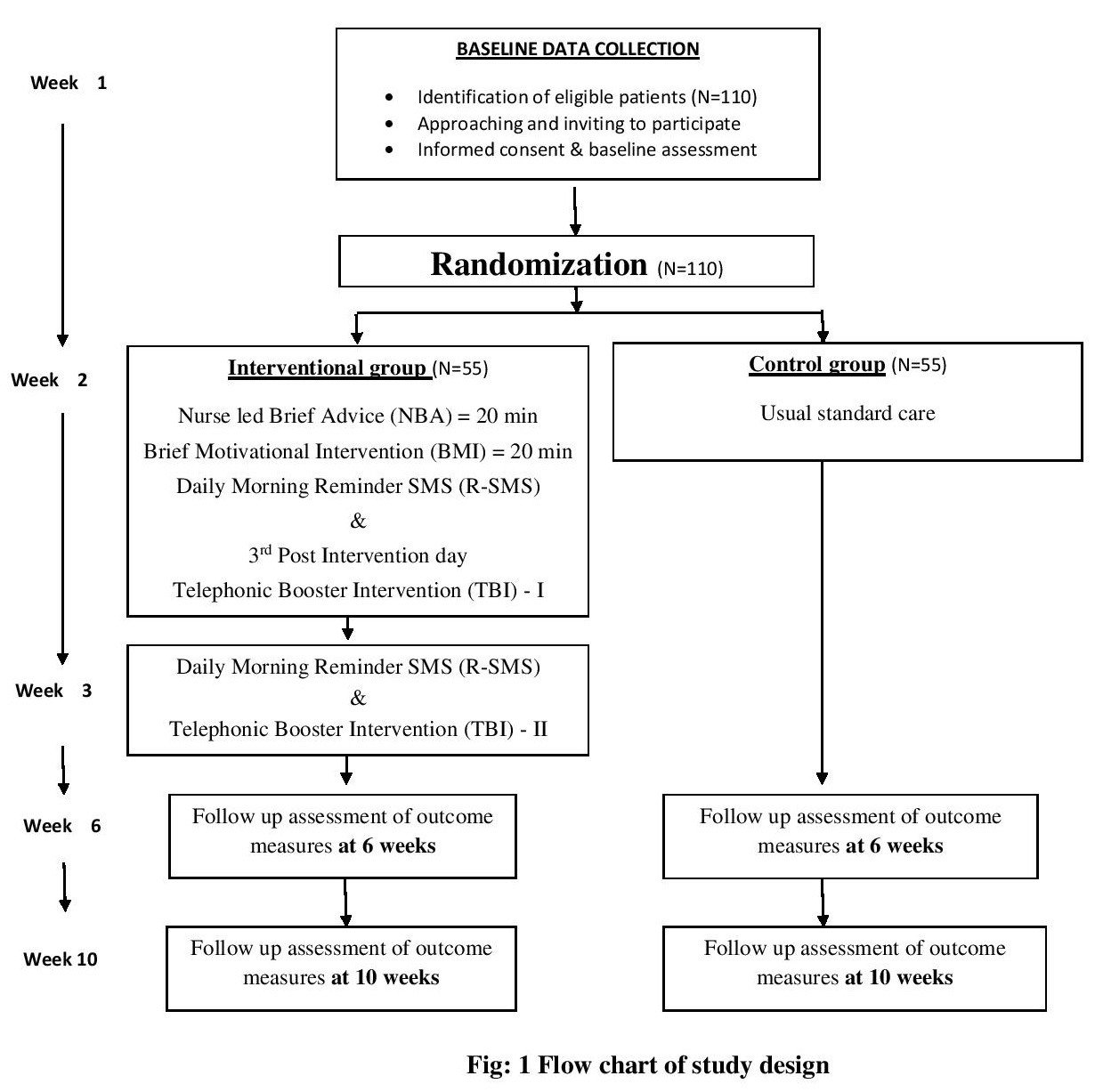
The study will adopt quantitative approach and the design will be a parallel arm block randomized controlled trial with 6- and 10-weeks follow-ups. The study design is guided by the CONSORT statement (Figure 1).
Study population & participants
CKD patients who are registered and receiving maintenance hemodialysis from selected hospital of Kerala, India will be study population. CKD patients, receiving hemodialysis, who fulfill inclusion and exclusion criteria, will be enrolled for the study. Baseline data will be collected after screening the participants and they will be randomized into an experimental or control group. The experimental group will receive nurse led interventions and the control group receives the standardized care. It will be voluntary for the patients to participate in the study.
Inclusion criteria will be adopted are patients:
-
To have a medical diagnosis of CKD and undergoing maintenance hemodialysis
-
To be aged above 20 years
-
Receiving hemodialysis at least for last three consecutive months
-
To have a mobile connection
-
To be able to read and understand Malayalam/Tamil/English
- The exclusion criteria will be:
-
Not willing to participate in intervention
-
A diagnosis of functional psychosis or organic brain disorder
-
Impaired cognition
-
Major visual or hearing impairments, or other sensory or motor impairments that may prohibit completion of the scheduled assessments
-
Limited life expectancy
Nurse led interventions (NLI)
This intervention is basically a process of empowering patients to make choices and lifestyle changes in line with treatment recommendations through the use of problem solving, goal setting and feedback. It emphasizes patients’ central role and responsibility in managing their illness. The program offers the potential for people to learn about their condition and treatment in a psychologically motivating and confidence enhancing structure.
Sessions will be facilitated by the researcher himself and the intervention components will include problem solving, overcoming barriers, challenging beliefs, conducting brainstorming sessions, goal setting, and reinforcement. Targeted behaviors will include fluid control and diet restriction. This nurse led interventions consist of nurse led brief advice, brief motivational intervention, telephonic booster intervention and daily morning reminder SMS.
-
Nurse led Brief Advice (NBA): Brief advice to dietary and fluid restriction includes information regarding guidelines for diet and fluid management in dialysis, its importance and consequences of non-adherence. Brief advice will be lasted for 20 minutes.
-
Brief Motivational Intervention (BMI): BMI is based on motivational interviewing, which will enhance the transformation of non-adherent behavior in to adherent behavior. The primary components consist of acknowledging the patient’s autonomy regarding their adherence behavior; providing personalized feedback based on preliminary assessment, encouraging patients to explore the pros and cons of dietary and fluid restriction; assessing importance, confidence and readiness to be adherent; supporting patients’ self-efficacy; and reinforcing efforts or intentions to make positive changes in behavior. Brief motivational interventions lasted an average of 20 minutes.
-
Telephonic Booster Intervention (TBI) & Daily Morning Reminder SMS (R-SMS): Telephonic booster intervention will be administered on the third post intervention day for about 10 minutes. During which the researcher will further identify the existing problems of patient based on feedback information. The researcher will utilize the situation to motivate the client to be adherent with the guidelines focusing on his/her strength and weakness with a purpose to ensure self efficacy and a change in behavior. From the second day of intervention, a daily reminder SMS will be sending to the participant’s mobile number until the 10th week.
Outcome measures and data collection
Primary outcomes are self reported adherence and biological & biochemical measures of adherence (inter-dialytic weight gain, serum potassium, phosphorus, urea, creatinine and blood pressure). Secondary outcomes are anxiety and depression, quality of life, self efficacy and dialysis and CKD related morbidities. These endpoints will be ascertained at baseline and at 6th and 10th week post-intervention by an independent assessor, who is not aware about the allocation.
The senior nurse in the dialysis center, will screen patients for eligibility and give a list of eligible patients. After baseline assessment, each participant will be randomized to either a control or intervention group. Three separate helpers will be there for generating sequence, enrollment and for allocation. This concealed randomization ensures both the researcher and participants are not aware of whether the next eligible participant will be allocated in to control or intervention arms. This will be masked until the time when participants are ready to receive intervention. Intervention will be administered by the researcher himself. Structured Interview technique and self reporting will be employed throughout the data collection period. Data will be collected during the hemodialysis session. Follow up measurements of outcomes will be assessed at 6th and 10th week in both groups.
Sampling technique and size
Purposive sampling technique will be adopted for the study. Eligible CKD patients will be sequentially recruited to the study. A sample size of 90 was estimated with the help of Openepi software15 with 80% power and 5% significance level to achieve the maximum effect at 80%. After estimating 20% attrition rate, the sample size is re-estimated into 110 (55 in each group).
Randomization and blinding
Following baseline measurement, each participant will be randomized to either a control or intervention group. Participants will be randomized by means of sequentially numbered opaque sealed envelopes (SNOSE) prepared according to a computer-generated random allocation order. Block randomization using randomly varying block sizes guarantee equal numbers of participants in each group with 1:1 allocation ratio. The blinding of participants and investigator is not possible here, however, the outcomes will be ascertained by independent observer (trained nurse) who will be unaware of the allocation and treatment arms of participants.
Ethical considerations/clearance
Institutional ethical committee permission has been taken. A study information sheet will be given to participants and the researcher will further verbally explain the study protocol and prerequisites in a simple language that patient can readily understand. Participation in the study will be of purely voluntary basis and will inform that they can withdraw at any time as they wish. A written informed consent shall be obtained from the participants before enrolling them in to the trial.
Plan for data analysis
After the data collection, descriptive and inferential statistics will be used for the data analysis. A statistical significance of 0.05 (P<0.05) will be adopted throughout the study.
DISCUSSION
Patient empowerment is the corner stone of the management of any chronic diseases. In such cases, adaptation with the treatment regimen and certain life style modifications are mandatory. As a well-trained health care professional nurses have tremendous role in the empowerment of such patients. Nursing care is always focused on multidimensional aspects of human being; they can influence the patient to ensure the necessary modifications in their daily life.
This theory based Nurse led intervention trial designed based on vigorous review of previous research emphasizing the importance of nurse specific interventions to empower patients and enable them to be adherent with their treatment regimen. Inclusion of both subjective and objective variables of adherence will enhance the strength of this trial and allows us to assess the broader impact on the intervention including both hard end points as well as patient reported outcomes. If the nurse led intervention is found to be effective, this cost effective potential program can be implemented in all dialysis centers by giving dialysis specific and psychologically imbued training to all dialysis nurses of our country that will really cater the needs of dialysis patients. The design of this trial will probably tackle limitations of prior studies by recruiting adequately sized and representative sample to determine clinically significant changes in biochemical markers, a theory based intervention protocol and careful measurement of both subjective and clinical variables at different follow up periods. Keenly designed, this trial will hopefully contribute to the wisdom of research, clinical practice and ultimately for the benefit of hemodialysis patients.
CONCLUSIONS
This study has started with a question, whether nurse lead interventions improve adherence to dietary and fluid restrictions and other health related outcomes among patients with chronic kidney disease? This theory based interventions and well driven trial will definitely detect a solid solution for this research question.
Acknowledgements
The authors would like to acknowledge Dr. Manju Thampi (Senior Nephrologist, NIMS), Dr. Manju Dhandapani (Senior faculty, PGIMER Chandigarh), Dr. Asha Sharma (Former INC Secretary), Mr. Shine Stephen (Faculty AIIMS) for their continuous support throughout the planning phase of this protocol.
Funding
This study had no financial support from any individuals or organizations.
Conflict of interest
The authors completed the Unified Competing Interest form at http://www.icmje.org/coi_disclosure.pdf (available upon request from the corresponding author) and declare no conflicts of interest.